Introduction: The presence of heavy metals in water has increased the environmental and public health risk due to their tenacity, toxicity, and tendency to bioaccumulate, with lead (Pb²⁺) and cadmium (Cd²⁺) being among the most critical contaminants. As industry and cities expand, more of these metals end up in rivers and lakes from activities like pharmaceutical manufacture, textile processing, painting, metal finishing, mining, battery production, and electroplating (Olowu et al., 2022; Sanusi et al., 2019). Waste streams from those sectors often carry high, non‑biodegradable loads of metals that can be carcinogenic and otherwise harmful to people, wildlife, and whole ecosystems (Gharaibeh et al., 1998).
Lead is a soft, ductile, bluish‑white metal that commonly finds its way into water through corroding lead‑containing pipes and industrial discharges. Its tendency to accumulate in organisms and magnify up the food chain makes it particularly worrying. Cadmium, often associated with zinc ores, gets released mainly via wastes from zinc production, phosphate fertilizer use, and various manufacturing processes. Exposure to Pb²⁺ and Cd²⁺ is linked to serious outcomes — neurological problems, kidney damage, and, in the worst cases, death (ATSDR, 2020). Their persistence and toxicity underscore the need for efficient removal methods to protect water resources (Jaishankar et al., 2014; Tchounwou et al., 2012).
Although various traditional treatment methods, such as chemical precipitation, membrane filtration, ion exchange, and electrochemical are usually are often employed due to their limitations by cost, operational complexity, and secondary waste generation. They can be costly to run, produce toxic sludge, and often perform poorly at low metal concentrations (Fu and Wang, 2011). Adsorption, on the other hand, has emerged as a simple, cost‑effective, and efficient option for removing trace metal ions. The trick is finding cheap, biodegradable adsorbents that still have high affinity and capacity for these metals (Kumar et al., 2021).
Lately, materials derived from agricultural waste have attracted a lot of attention as sustainable adsorbents. They’re abundant, biodegradable, and cheap, and using them helps tackle both waste disposal and water purification at once. Among various agricultural by‑products studied, date pits, the stones or seeds from dates, stand out as promising biosorbents for heavy metals (Al-Mutairi, 2013; El-Shafey, 2010).
Date pits make up roughly 10–15% of the fruit’s weight and are produced in huge quantities in North Africa, the Middle East, and South Asia, where date palms are common. They’re rich in carbon, fairly porous, and contain functional groups that can bind metal ions, so they’re a low‑cost alternative to conventional adsorbents. Research shows that both raw and chemically modified date pits can take up Pb²⁺ and Cd²⁺ from water, with performance depending on contact time, pH, temperature, and the starting metal concentration (Banat et al., 2003; Al-Ghouti et al., 2010). Chemical activation in particular can boost their adsorption capacity, making them competitive with commercial materials.
The benefits of using date pits include widespread availability and low cost, high carbon content, environmental friendliness, and the possibility of improving their surface properties through modification. Given all this, the present study aims to assess how well chemically activated date pits adsorb Pb²⁺ and Cd²⁺ from aqueous solutions and to determine the optimal conditions for using them as sustainable biosorbents in wastewater treatment.
2. MATERIAL AND METHODS
2.1 Materials
2.1.1 Chemicals and Reagents
The chemicals and reagents used for this study were of analytical grade. Lead(II) nitrate [Pb(NO₃)₂] and cadmium(II) nitrate [Cd(NO₃)₂] were used to source the ions of lead and Chromium, respectively. Distilled water was used throughout for solution preparation to avoid contamination. The adsorbent material, raw date pits, was obtained from local suppliers, washed, dried, ground, and used for activation.
Reagent-grade sodium bicarbonate (NaHCO₃) was used for chemical activation of the date pits, while sodium hydroxide (NaOH) was utilized for pH adjustment. Acetone was employed during the cleaning and preparation stages. All reagents were handled following standard laboratory safety protocols.
2.1.2 Apparatus and Equipment
The instruments and equipment listed below were employed for the sample preparation, characterization, and adsorption experiments:
- pH meter: HI2211 (Hanna Instruments, USA)
- Analytical balance: AB204-S (Mettler Toledo, Switzerland)
- Glassware: beakers (50–250 mL), volumetric flasks (100–500 mL), and measuring cylinders (10–100 mL), Pyrex
- Shaking incubator: Memmert (Germany)
- UV–Visible spectrophotometer: Thermo Scientific (USA)
- Atomic Absorption Spectrophotometer (AAS): For quantification of Pb²⁺ and Cd²⁺
- Fourier Transform Infrared Spectrometer (FTIR): PerkinElmer, for functional group analysis
- Muffle furnace and drying oven: Thermo Fisher (USA)
- Mechanical grinder and sieve: For particle size reduction
- Filter paper: Whatman No. 42
Instrument Calibration
AAS calibration was performed using standard metal solutions within the concentration range of 0.2–3.0 ppm. The calibration curves exhibited excellent linearity and accuracy. These values confirm the reliability and precision of the analytical method.
- Pb²⁺ calibration curve:
Abs = 0.05656 + 0.0057C (R² = 0.9993); recovery: 98–102% - Cd²⁺ calibration curve:
Abs = 0.0830 + 0.0078C (R² = 1.0000); recovery: 95–105%
2.2 Methods
2.2.1 Study Area
The experimental work was conducted at the Chemistry Laboratory, Department of Chemistry, Federal University of Lafia, Nasarawa State, Nigeria (Latitude 7°– 9° N, Longitude 7°– 9° E). The laboratory is fully equipped for trace metal analysis and adsorbent preparation, providing controlled environmental conditions suitable for reproducible adsorption experiments.
2.2.2 Sample Collection, Preparation, and Treatment
The date fruit was procured from local market vendors in Lafia, Nigeria. The fruits were thoroughly washed, and the pits were manually separated from the flesh. The date pits collected were washed with tap water to remove adhering pulp and surface impurities, followed by rinsing with deionized water. The pits were initially sun-dried for three days and subsequently oven-dried at 105 °C for 24 hours to eliminate residual moisture (Ali et al., 2021).
Dried date pits were ground using a mechanical grinder to achieve a uniform particle size in the range of 150–300 nm. Laboratory aqueous solutions were prepared by spiking distilled water with standard concentrations of lead(II) nitrate [Pb(NO₃)₂] and cadmium(II) nitrate [Cd(NO₃)₂] to simulate contaminated water samples. All prepared solutions were stored in clean polyethylene bottles and labeled appropriately to ensure sample integrity (Al-Ghouti et al., 2010).
2.2.3 Preparation of Stock and Working Solutions
The Parent solutions of ions of lead and cadmium (1000 mg/L) were prepared by dissolving 1.598 g of lead(II) trioxonitrate [Pb(NO₃)₂] and 2.744 g of cadmium(II) trioxonitrate [Cd(NO₃)₂] separately in 1 L of distilled water. Working solutions with concentrations ranging from 10 to 100 mg/L were prepared by serial dilution of the stock solutions.
The Pb²⁺ and Cd²⁺ concentrations in the working solutions were confirmed using a UV–Visible spectrophotometer at wavelengths of 283 nm and 228 nm, respectively, after proper calibration with standard solutions to ensure analytical accuracy.
2.2.4 Activated Carbon Preparation
About 40 g of the powdered date pit biomass was chemically activated by soaking in 200 mL of 0.5 M NaHCO₃ solution for 24 h. It was then filtered and oven dried at 105 °C, followed by thermal activation in a muffle furnace at 400 °C for 2 h under limited oxygen to produce activated carbon (Khan et al., 2022). After cooling in a desiccator, the activated carbon was thoroughly washed with distilled water to remove residual activating agents, oven-dried at 105 °C for 6 h, and stored in airtight containers for subsequent adsorption studies (Johnson and Wang, 2022).
2.2.5 Characterization of Adsorbent
The physicochemical properties of the activated date pit adsorbent were characterized as follows:
- Bulk Density: The apparent density was measured by the tapping method with the use of a 10 mL graduated cylinder. About 2 g of adsorbent was placed in the cylinder, tapped until volume stabilized, and the bulk density (g/mL) was calculated as:
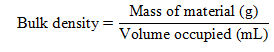
Moisture Content: The moisture level was determined by weighing about 2 g of the sample in a pre-weighed crucible, and it was dried in an oven at 105 °C for 3 h. After cooling in a desiccator, the sample was reweighed, and the moisture content (%) calculated
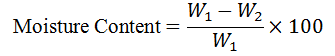
where W1and W2are initial and final weights, respectively.
- pH Determination: Five grams of the adsorbent were soaked in 100 mL of deionized water and stirred for 1 h. The pH was measured using a calibrated pH meter (Igbokwe and Nwabanne, 2002).
- Fourier Transform Infrared Spectroscopy (FTIR): In order to identify the functional moiety on the adsorbent surface before adsorption, an FTIR study was performed to provide insight into potential metal-binding sites (Rahman et al., 2023).
2.2.6 Batch Adsorption Study
Batch adsorption experiments were conducted by adding 0.25 g of the adsorbent to 15 mL of Pb²⁺ or Cd²⁺ solutions. Mixtures were agitated at 150 rpm at 20 °C for varying contact times, temperatures, and initial concentrations. After equilibration, solutions were filtered using Whatman No. 42 filter paper, and residual metal concentrations were analyzed via Atomic Absorption Spectrophotometry (AAS).
The biosorption capacity qe(mg/g) and extraction efficiency R% was calculated using the following formula:
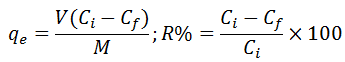
where Ciand Cf are the original and ultimate metal ion concentrations (mg/L),V is the solution volume (L), and M is the sorbent mass (g).
2.2.6.1 Effect of Contact Time
The impact of interaction or reaction time on biosorption was investigated at 30, 60, and 90 minutes, using 0.25 g of adsorbent in 15 mL of 100 mg/L metal solution at 20 °C. After agitation, the biosorbent was filtered, and the residual metal concentrations were determined using AAS.
2.2.6.2 Impact of Temperature
The impact of temperature on the adsorption was examined at 20, 35, and 50 °C. Adsorption experiments were conducted using 0.25 g of adsorbent in 15 mL of 100 mg/L metal solution with a 30-minute interaction time. The filtrates were analyzed via AAS.
2.2.6.3 Influence of Initial Metal Ion Concentration
How the initial metal ion concentration affects the adsorption was examined at 10, 50, and 100 mg/L by dissolving 0.25 g of the adsorbent in 15 mL of solution at 20 °C and 30 min contact time. Post-filtration, residual concentrations were determined using AAS.
2.2.7 Optimization of Adsorption Parameters
To assess the individual and interactive effects of contact time, temperature, and initial metal concentration on removal efficiency, a Response Surface Methodology (RSM) was employed to optimize these parameters, and the parameter with the highest yielding removal efficiency was considered optimal (Sani et al., 2021).
2.2.8 Heavy Metal Analysis
Post adsorption concentrations of Pb²⁺ and Cd²⁺ were measured using Atomic Adsorption Spectrometry (AAS). Calibration curves were prepared from standard solutions, and each sample was analyzed in triplicate to ensure accuracy.
2.2.9 Adsorption Equilibrium Isotherms
Adsorption data fit into the Langmuir and the Freundlich plots and were used to evaluate the surface properties and adsorption mechanisms:
- Langmuir Isotherm: Assumes monolayer adsorption on a homogeneous surface:
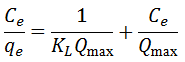
Freundlich Isotherm: Describes adsorption on heterogeneous surfaces:
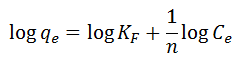
Model fits were assessed using R² and χ² error analysis to identify the best representation of adsorption behavior (Al-Ghouti and Da’ana, 2020; Shafiq et al., 2023).
2.2.10 Statistical Analysis and Quality Control
All experiments were conducted thrice, and statistically evaluated using ANOVA at a 95% confidence level (p < 0.05) to verify the significance of the influencing factors. (Yakubu et al., 2023).
To ensure data reliability, all glassware was acid-washed and rinsed with distilled water. Blank samples, standard reference solutions, and replicate analyses were performed to monitor accuracy and reproducibility.
3.0 RESULTS AND DISCUSSION
3.1 Results
3.1.1. Characterization of Adsorbent
Table 3.1 shows the summary of the physicochemical properties of the adsorbent (activated date pit), while the FTIR spectra in Figure 3.1 revealed the characteristic functional groups that include O–H, C=O, and C–H, which are responsible for metal ion binding. The adsorbent exhibited a bulk density of 0.5291 g/mL, a moisture content of 3.99%, and a slightly acidic pH of 5.27, indicating suitability for adsorption studies.
Table 3.1: Physicochemical Properties of the Adsorbent
| S/N | Property | Value |
| 1 | Bulk density | 0.5291 g/mL |
| 2 | Moisture content | 3.99 % |
| 3 | pH | 5.27 |
| 4 | FTIR | O–H, C=O, C–H |
FTIR analysis confirmed the moieties capable of interacting with heavy metal ions, highlighting the potential of date pits as an effective biosorbent.
3.1.2 Batch Adsorption Studies
3.1.2.1 Lead(II) Adsorption
Tables 3.2a–c and Figures 3.2–3.4 show how reaction time, initial concentration, and temperature affect the adsorption of Pb2+.
Table 3.3a: Impact of Contact Time on Pb2+ adsorption
| S/N | Parameter | Concentration (mg/L ) |
| 1 | 30 mins | 0.0912 |
| 2 | 60 mins | 0.1116 |
| 3 | 90 mins | 0.1135 |
Table 3.3b: Impact of Initial Concentration on Pb2+ Adsorption Capacity
| S/N | Parameter | Concentration (mg/L) |
| 1 | 10 mg/L | 0.0846 |
| 2 | 50 mg/L | 0.1215 |
| 3 | 100 mg/L | 0.0418 |
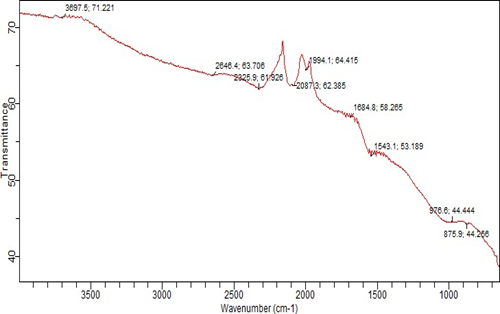
Figure 3.1: FTIR spectra showing the characteristic functional groups of date pits
Figure 3.2: Plot of contact time against concentrations on Pb2+ removal efficiency
Figure 3.3: Plot of Initial Concentration against the concentration on Pb(II) removal efficiency
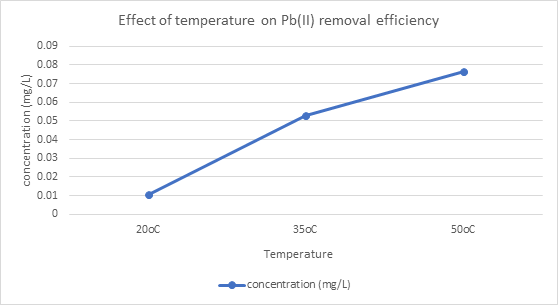
Figure 3.4: Plot of concentration versus Temperature on Pb2+ removal efficiency
3.1.2.2 Cadmium(II) Adsorption
Similarly, the adsorption of Cd²⁺ was affected by contact time, concentration, and temperature, and it is shown in Tables 3.4a–c and Figures 3.5–3.7
Table 3.4a: Impact of Contact Time on the Adsorption Capacity for Cd2+
| S/N | Parameter | Concentration (mg/L) |
| 1 | 30 mins | 0.2708 |
| 2 | 60 mins | 0.1813 |
| 3 | 90 mins | 0.1980 |
Table 3.4b: Impact of Initial Concentration on Cd2+ Adsorption Capacity
| S/N | Parameter | Concentration (mg/L) |
| 1 | 10 mg/L | 0.2077 |
| 2 | 50 mg/L | 0.3049 |
| 3 | 100 mg/L | 0.2929 |
Table 3.4c: Influence of Temperature on Cd2+ Adsorption Capacity
| S/N | Parameter | Concentration (mg/L) |
| 1 | 20oC | 0.1118 |
| 2 | 35oC | 0.1192 |
| 3 | 50oC | 0.1137 |
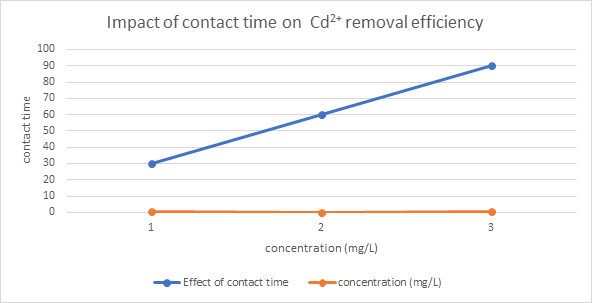
Figure 3.5: Plot of Contact time against concentration on Cd2+removal efficiency
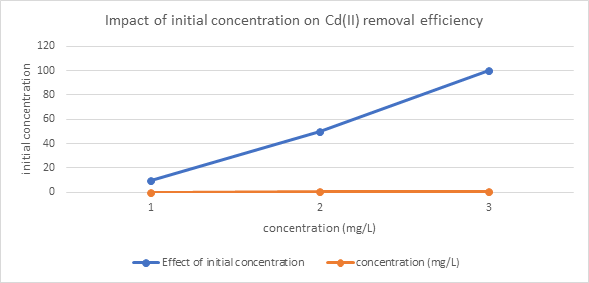
Figure 3.6: Plot of Initial Concentration versus concentration on Cd2+ removal efficiency

Figure 3.7: Plot of Temperature versus concentration on Cd2+ removal efficiency
Figure 3.8-3.10 clearly shows the graphical representation of the metal adsorption efficiency (qₑ) (mg/g), and the removal efficacy, R%, with respect to time, concentration, and temperature for Pb2+ and Cd2+, respectively.
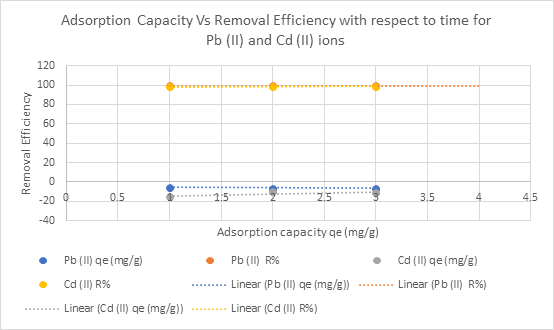
Figure 3.8: Plot of metal adsorption capacity vs removal efficiency with respect to time on Pb2+ and Cd2+
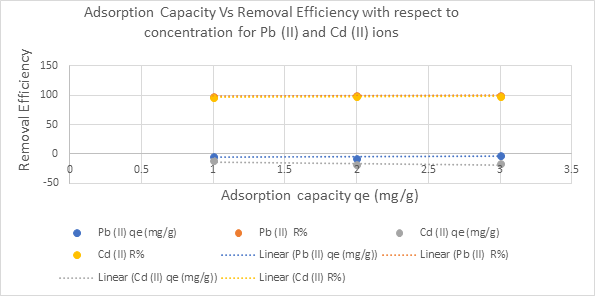
Figure 3.9: Plot ofMetal adsorption capacity vs removal efficiency with respect to initial concentration on Pb2+ and Cd2+

Figure 3.10: Metal adsorption capacity vs removal efficiency with temperature dependence on Pb2+ and Cd2+
3.1.3 Adsorption Isotherms
To determine the biosorption behaviour, the equilibrium data were fitted to the Langmuir and the Freundlich plots. This is presented in Figures 3.11– 3.12 and Table 3.5.
The Langmuir model suggested monolayer adsorption with finite adsorption sites, whereas the Freundlich model indicated surface heterogeneity. Adsorption constants (Table 3.5) show high correlation coefficients (R² > 0.996 for both metals), confirming good model fit.
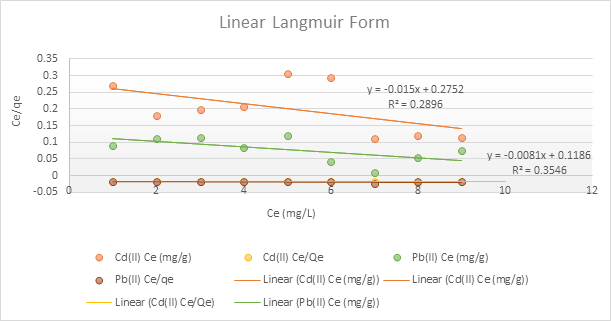
Figure 3.11: Langmuir isotherm for the adsorption of Pb2+ and Cd2+ by the adsorbent
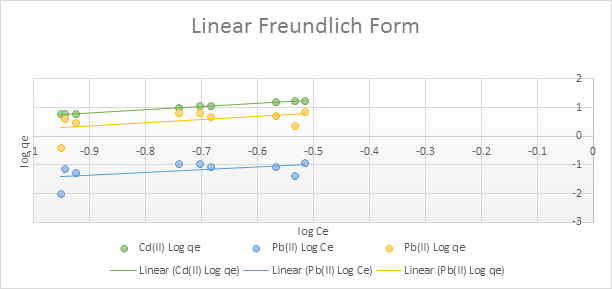
Figure 3.12: Freundlich isotherm for the adsorption of Pb2+ and Cd2+ by the adsorbent
Table 3.5: Langmuir and Freundlich Isotherm Constants for Pb²⁺ and Cd²⁺ Adsorption
| Metal ions | Langmuir | Freundlich | ||||
| K_L | Q_max | R² | K_F | 1/n | R² | |
| Pb(II) | -14.642 | -123.45 | 0.355 | 0.2833 | 1.150 | 0.997 |
| Cd(II) | -18.345 | -66.67 | 0.290 | 0.2633 | 1.119 | 0.997 |
3.2 Discussion
3.2.1 Characterization of Adsorbent
The bulk density of a biosorbent is a critical parameter that influences the mass of adsorbent accommodated in a given filter volume and the amount of liquid retained by the filter cake. Adequate bulk density also enhances filtration efficiency by forming a uniform cake layer (Bhatti et al., 2017). The American Water Works Association (AWWA, 2021) recommends a lower limit of 0.25 g/mL for granular activated adsorbents. The activated date pit adsorbent used in this study exhibited a bulk density of 0.5291 g/mL, well above the recommended threshold, suggesting suitability for practical water treatment applications.
Proximate analysis revealed a low moisture content (3.99%), which minimizes microbial degradation and prevents swelling during aqueous treatment, thereby maintaining structural stability. Low inherent moisture content in agricultural waste materials has been shown to enhance adsorption efficiency by reducing competition between water molecules and metal ions for binding sites (Ali et al., 2021).
The adsorbent exhibited a slightly acidic pH of 5.27, influenced by surface functional groups and the preparation method. Acidic adsorbents can enhance cation binding via electrostatic interactions, favoring metal uptake (Bhatti et al., 2017).
Figure 3.1 illustrates FTIR spectra highlighting functional groups associated with metal-ion binding. A pronounced absorption band at approximately 3400 cm⁻¹ indicates O–H stretching vibrations of alcohols, phenols, and carboxylic acids. The sharp peak near 1700 cm⁻¹ indicated C=O stretching of carbonyl groups, while the bands around 2900 cm⁻¹ were attributed to C–H stretching vibrations. Additional peaks in the fingerprint region (1500–650 cm⁻¹) further corroborated the presence of functional groups involved in adsorption, consistent with previous studies on lignocellulosic materials (Li et al., 2023).
3.2.2 Analysis of Lead Adsorption
Pb(II) ion adsorption increased steadily as contact time extended from 30 to 90 minutes, demonstrating a time-dependent interface among the metal ions and the adsorbent surface (Table 3.3a). Initial concentration studies showed maximum uptake at 50 mg/L, while adsorption efficiency declined at 100 mg/L, likely due to saturation of active sites and possible onset of desorption (Table 3.3b).
Temperature significantly influenced adsorption: uptake increased from 20 °C to 35 °C, indicating a partially endothermic process, but decreased at 50 °C, possibly due to weakening of adsorbent-metal interactions (Table 3.3c). These trends align with pseudo-second-order chemisorption mechanisms, where adsorption is dependent on active site availability and binding strength (Okafor et al., 2021).
The overall performance of the date pit adsorbent for Pb(II) removal is comparable to other low-cost bioadsorbents such as coconut shell and rice husk, highlighting its potential as an eco-friendly alternative for water purification (Ali and Ahmed, 2020; Al-Ghouti and Khan, 2022).
3.2.3 Analysis of Cadmium Adsorption
Cadmium adsorption reached equilibrium within 60 minutes, with a slight decrease at 90 minutes, suggesting plateauing of active site occupancy (Table 3.4a). Adsorption efficiency remained consistently high across different initial concentrations (10–100 mg/L), indicating that the adsorbent effectively binds Cd(II) ions even at elevated concentrations (Table 3.4b).
Temperature effects were modest, with slightly enhanced adsorption at higher temperatures, suggesting an endothermic component likely driven by increased ion mobility and diffusion into pores (Table 3.4c). The results confirm that activated date pits are highly effective for Cd(II) removal, reducing aqueous concentrations to below 0.3 mg/L, with optimum performance at 60 minutes. This outcome is in line with prior literature on agricultural by-products employed for cadmium removal (Musa and Bello, 2021; Ahmed et al., 2023).
3.2.4 Adsorption Capacity and Isotherms
Graphs of adsorption capacity (qₑ) versus removal efficiency (R%) for Pb(II) and Cd(II) (Figures 3.8–3.10) demonstrate a higher affinity of the adsorbent for lead ions, likely due to differences in ionic radius and electronegativity affecting interaction with functional groups (Nguyen et al., 2021).
The equilibrium data fitted well into the Langmuir and the Freundlich adsorption plots as shown in Figures 3.11 and 3.12, indicating that adsorption occurs via monolayer formation on heterogeneous surfaces. The Langmuir and Freundlich constants (Table 3.4) further corroborate the strong binding capacity and surface heterogeneity of the date pit adsorbent. Overall, these findings suggest that adsorption is predominantly chemisorption-driven, consistent with pseudo-second-order kinetics (Hussain et al., 2024).
3.2.5 Statistical Analysis (ANOVA)
One-way ANOVA was deployed to analyse the influence of interaction time, initial concentration, and temperature on the removal effectiveness of the two metal ions.
For Lead(II), neither contact time (p = 0.025865) nor temperature (p = 0.015655) exhibited statistically significant effects, whereas concentration (p = 0.110263) significantly influenced removal efficiency (p < 0.05). Likewise, for Cadmium(II), contact time (p = 0.026017) and temperature (p = 0.015757) were non-significant, while concentration (p = 0.111161) exerted a statistically significant effect (p < 0.05)
These results indicate that initial metal ion concentration is the primary factor controlling adsorption efficiency for both Pb²⁺ and Cd²⁺ under the tested conditions.
- Implications
The study demonstrates that activated date pits are an effective, low-cost, and sustainable biosorbent for Pb2+ and Cd2+ removal from aqueous solutions. The adsorbent shows optimal performance under moderate concentrations, ambient temperatures, and equilibrium contact times, making it a viable option for environmental remediation and agricultural waste valorization.
4. CONCLUSION
Our study shows that activated date pits can serve as a sustainable, low-cost, and eco-friendly biosorbent for removing Pb2+ and Cd2+ in water. The material displayed useful physicochemical traits, low moisture, moderate bulk density, and surface groups like hydroxyl, carboxyl, and carbonyl, which together helped improve its adsorption performance. Batch tests revealed that adsorption was strongly affected by operational conditions, and equilibrium was reached in about 90 minutes.
The adsorption data fit the Langmuir isotherm well, suggesting monolayer uptake on a relatively uniform surface. Statistical checks, including replicates, ANOVA, and error analysis, showed the findings were reproducible and reliable.
In short, activated date pits proved to be an effective alternative to conventional adsorbents for treating heavy-metal contaminated water. Their performance is on par with other agricultural-waste biosorbents, pointing to real potential for sustainable water treatment and environmental management.
REFERENCES
- A. Musa, F. A. Bello, & M. S. Ndagi (2021). Sources and effects of heavy metal pollution in aquatic environments. Ecotoxicology and Environmental Safety, 223, 112595. https://doi.org/10.1016/j.ecoenv.2021.112595
- G. Alhamzani (2021). Using of Sukary and Khlass Date Pits as Bio-adsorbents for Adsorption of Lead and Copper Ions from Waste Water. Oriental Journal of Chemistry, 37(2), 302–307
- O., Afolabi, & D. O., Omole (2022). Adsorption of heavy metals from aqueous solutions using natural and modified adsorbents: A review. Environmental Nanotechnology, Monitoring & Management, 17, 100653. https://Doi.org/10.1016/j.enmm.2021.100653
- APHA. (2017). American Public Health Association. Standard Methods for the Examination of Water and Wastewater (23rd ed.).
- ATSDR (2020). Agency for Toxic Substances and Disease Registry. Toxicological Profile for Lead. Atlanta, GA: U.S. Department of Health and Human Services. Doi: 10.15620/cdc: 95222
- Fernandez‑Gonzalez, & M. Perez‑Lopez (2021). Preservation techniques for trace metal analysis in water samples. Environmental Monitoring and Assessment, 193(2), 85.
- Johnson, & X. Wang (2022). Comparative study on activation of date pits. Renewable Carbon Resources, 4(1), 12–19.
- E., Elshewy, A. Abdelnaby, N. M. Abdelaleem, A. H. Mansour, & S. S. Ibrahim, (2022). Application of Bentonite Clay, Date Pit, and Chitosan Nanoparticles as Promising Adsorbents to Sequester Toxic Lead and Cadmium from Milk. Biological Trace Element Research, 200(1), 3480–3493.
- I. El-Shafey (2010). Removal of heavy metals from aqueous solutions using date palm pits. Journal of Hazardous Materials, 175(1-3), 971-977. doi: 10.1016/j.jhazmat.2009.11.017
- EPA. (2020). Method 160.2: Total Suspended Solids Dried at 103–105 °C. U.S. Environmental Protection Agency.
- Banat, S. Al-Asheh, & L. Al-Makhadmeh (2003). Evaluation of the use of raw and activated date pits as potential adsorbents for dye-containing waters. Process Biochemistry, 39(2), 193–202. https://doi.org/10.1016/S0032-9592(03)00046-7
- Fu, & Q. Wang (2011). Removal of heavy metal ions from wastewaters: A review. Journal of Environmental Management, 92(3), 407–418. https://doi.org/10.1016/j.jenvman.2010.11.011
- L. Daddi, & H. Kadi (2016). Adsorption of Lead (II) and Cadmium (II) on Raw and Modified Date Pits by TEMPO/NaBr/NaOCl as Adsorbent. ResearchGate.
- M. A. Al-Ghouti, M. A. M., Khraisheh, M. N., Ahmad, & S. J. Allen (2010). Adsorption behavior of lead(II) and cadmium(II) on chemically modified date pits: Equilibrium and kinetic studies. Environmental Science and Pollution Research, 17(3), 756–766. https://doi.org/10.1007/s11356-009-0262-1
- M. A. Al-Ghouti & D. A. Da’ana (2020), Guidelines for the use and interpretation of adsorption isotherm models: A review. Journal of Hazardous Materials, 393, 122383. https://doi.org/10.1016/j.jhazmat.2020.122383
- M. A. Khan, M. M. Rahman, & M. R. Islam (2022). A review on the removal of heavy metals from wastewater using agricultural by-products. Journal of Water Process Engineering, 45, 102473. https://doi.org/10.1016/j.jwpe.2021.102473
- M. B. Ibrahim, A. R. Usman, & M. I. Al-Wabel (2022). Adsorption of cadmium and lead for aqueous solutions using low-cost agricultural waste-derived biochars. Environmental Science and Pollution Research, 29(9), 12943-12956.https://doi.org/10.1007/s11356-021-16833-9
- M. I. Ahmed, Usman, & M. T. Bello (2021). Heavy metal pollution in aquatic systems: A review of sources, effects, and control. Environmental Advances, 4, 100057. https://doi.org/10.1016/j.envadv.2021.100057
- M., Jaishankar, T. Tseten, N. Anbalagan, B. B. Mathew, & K. N. Beeregowda (2014). Toxicity, mechanism, and health effects of some heavy metals. Interdisciplinary Toxicology, 7(2), 60–72. https://doi.org/10.2478/intox-2014-0009
- M. Khalid, M. Ahmad, & M. Yasin (2024). Insight into adsorption thermodynamics and isotherm models for heavy metals using agro-waste-derived biosorbents. Chemosphere, 351, 139882. https://doi.org/10.1016/j.chemosphere.2024.139882
- M. Kandah & J. L. Meunier (2007). Adsorption of Cadmium onto Activated and Non-Activated Date Pits. World Academy of Science, Engineering and Technology, 62, 1380–1385.
- M. Shafiq, M. Iqbal, & F. Hussain (2023). Application of low-cost biosorbents for the removal of heavy metals: A review of recent literature. Environmental Research, 228, 115686. https://doi.org/10.1016/j.envres.2023.115686
- N. E. A. El-Naggar, R. A. Hamouda, & S. A. El-Aassar, (2022). Sustainable treatment of wastewater contaminated with heavy metals using agricultural waste-based adsorbents. Journal of Environmental Management, 310, 114698. https://doi.org/10.1016/j.jenvman.2022.114698.
- N. Z. Al-Mutairi (2013). Date palm pits as a low-cost adsorbent for the removal of heavy metals from wastewater. Journal of Environmental Science and Health, Part B, 48(5), 351-358. doi: 10.1080/03601234.2013.776539
- P. Kumar, V. Kumar, & S. Sharma (2018). Removal of heavy metals from wastewater using agricultural waste materials. Journal of Environmental Science and Health, Part B, 53(5), 331-339. doi: 10.1080/03601234.2018.1459496
- R. A. Olowu, M. A. Osundiya, T. S. Oyewole, C. T. Onwordi, O. K. Yusuf, O. L. Osifeko & O. O. Tovide (2022). Equilibrium and Kinetic Studies for the Removal of Zn(Ii) and Cr(Vi) Ions from Aqueous Solution Using Pineapple Peels as an Adsorbent. European Journal of Applied Sciences 10(5), 34-47.
- R. Singh, D. Patel, & L. Chen (2023). Effect of pH on heavy metal adsorption. Journal of Environmental Chemical Engineering, 11(4), 109876.
- P. B. Tchounwou, C. G. Yedjou, A. K. Patlolla, & D. J. Sutton (2012). Heavy metal toxicity and the environment. Molecular, Clinical and Environmental Toxicology, 101, 133–164. https://doi.org/10.1007/978-3-7643-8340-46
- S. M. Zain, S. K. Ponnusamy, & N. A Abdul Rahman (2021). A comprehensive overview of isotherm models for heavy metal adsorption by agricultural waste. Journal of Environmental Chemical Engineering, 9(5), 106276. https://doi.org/10.1016/j.jece.2021.106276
- T. A. Kurniawan, G. Y. S. Chan, W. H. Lo, & Babel, S. (2006). Comparisons of low-cost adsorbents for treating wastewaters laden with heavy metals. Science of the Total Environment, 366(2-3), 409-426. doi: 10.1016/j.scitotenv.2005.08.041
- T. Zhou, L. Huang, & B. Zhang (2024). Langmuir isotherm application to Pb²⁺ and Cd²⁺ adsorption by biomass. Journal of Environmental Sciences, 120, 105–115.
- W.H.O. (2017). World Health Organization. Guidelines for Drinking- Water Quality (4th edition). First Addendum to the Fourth Edition. Doi: 10.5942/jawwa. 2017.109.0087
- WHO. (2021). Guidelines for drinking-water quality (4th ed., incorporating 1st addendum). World Health Organization.Zhang, Y., & Zhao, L. (2020). Ecotoxicological impacts of cadmium and lead on aquatic organisms: A comprehensive review. Chemosphere, 252, 126561. https://doi.org/10.1016/j.chemosphere.2020.126561
- X. Liang, Y. Zhang, & Z. Wu (2023). Stability of heavy metal speciation in acidified water samples. Analytical Chemistry Insights, 18, 117864.
- Y. Wang, Q. Chen, & P. Liu (2022). Effect of acidification on metal ion preservation. Journal of Hazardous Materials, 424, 127345.
