Introduction
Nanoparticles in chemistry are relatively new area of nanoscience. Nanoparticles have always been in existence in nature, in form of smoke and dust, many chemist still find these statements perplexing even thirty years ago [1]; [2]. Nanoparticles and its properties have been used long ago, and were discovered in stained glass, paints, and building materials. Transition metal nanoparticles have been employed extensively as heterogeneous catalysts for more than a century, producing significant profits for petrochemical industries [3]; [4]. Despite these ubiquitous instances, it wasn’t until the end of the 20th century that nanoparticle chemistry developed into a rigorous academic discipline due to the development of electron microscopy and other contemporary characterization techniques, which gave researchers the ability to analyse object sizes as small as nanometers [5]. The scientific community realized the need for innovative chemistries for nanoparticle synthesis, purification, and post-synthetic alterations because many basic studies and technology applications required nanoparticles with consistent sizes and shapes. Numerous techniques can be employed to create nanoparticles although; they are commonly supplied as dispersions in different media, including glasses or liquid solvents, and can also be produced in the gas phase to create aerosols [6]. The Majority of the current research has been on creating nanoparticles via colloidal approaches employing polar and non-polar solvents as the reaction medium. It found out that solution-phase syntheses are the most practical and adaptable [3]. A thorough understanding of the nucleation and growth processes was necessary to master the synthesis of nanoparticles, and this led to active research in adjacent fields of materials characterization and modeling. There are still many fascinating discoveries to be made therefore this effort is far from finished. Readers are informed about the most recent advancements in nanoparticle chemistry via this special edition of Chemical Reviews [7].
“Nanotechnology” describes a young branch of research that deals with the creation and synthesis of diverse nanomaterials with particle sizes between one and one hundred nm that may differ from the bulk material due to their size are known as nanoparticles [8]; [9]. Current methods for producing various metallic nanomaterials include the use of copper, zinc, titanium, magnesium, gold, alginate, and silver [10]. Nanoparticles are being employed for diverse objectives, from medical treatments, using in numerous areas of industry production such as solar and oxide fuel batteries for energy storage, to wide incorporation into diverse materials of everyday usage such as cosmetics or garment [5].
Nanoparticles are generally classified into three; Organic Nanoparticles: Dendrimers, micelles liposomes, ferritin. Inorganic Nanoparticles: This is sub divided into two:
i Metal-based nanoparticles: Aluminum (Al), Cadmium (Cd), Cobalt (Co), Gold (Ag) Iron (Fe), Lead (Pb), Silver (Au), Zinc (Zn) [7].
ii Metal-oxide based nanoparticles: Aluminum oxide, Cerium oxide, Iron oxide, Magnetite, Silicon dioxide, Titanium oxide, Zinc oxide [11].
Carbon based nanoparticles: Fullerenes, graphene, carbon nanotubes (CNT), carbon nanofibers, carbon black [11].
- Cocos nucifera is a large palm, growing up to 30 metres (100 feet) tall, with pinnate leaves 4 – 6 m (13 – 20 ft) long, and pinnae 60 – 90 centimetres (2 – 3 ft) long; old leaves break away cleanly, leaving the trunk smooth. On fertile soil, a tall coconut palm tree can yield up to 75 fruits per year, but more often yields less than 30. Given proper care and growing conditions, coconut palms produce their first fruit in six to ten years, taking 15 to 20 years to reach.
- Peak production.

Figure 1: Orange Tree and Fruit [12], Palm Tree [13] and Well Labeled Diagram of Coconut fruit [14].
Botanically, the coconut fruit is a drupe, not a true nut like other fruits and it consist of three layers: the exocarp, mesocarp, and endocarp [15]. The exocarp is the glossy outer skin, usually yellow-green to yellow-brown in color. The mesocarp is composed of a fiber, called coir, which has many traditional and commercial uses [16] . Both the exocarp and the mesocarp make up the “husk” of the coconut, while the endocarp makes up the hard coconut “shell”. The endocarp is around 4 millimetres (1⁄8 inch) thick and has three distinctive germination pores (micropyles) on the distal end. Two of the pores are plugged (the “eyes”), while one is functional. [17]; [15]. The interior of the endocarp is hollow and is lined with a thin brown seed coat around 0.2 mm (1 ⁄ 64 in) thick. The endocarp is initially filled with a multinucleate liquid endosperm (the coconut water). As development continues, the cellular layers of endosperm is deposit along the walls of the endocarp up to 11 mm (3 ⁄ 8 in) thick, starting at the distal end. They eventually form the edible solid endosperm (the “coconut meat” or “coconut flesh”) which hardens over time. The small cylindrical embryo is embedded in the solid endosperm directly below the functional pore of the endosperm. During germination, the embryo pushes out of the functional pore and forms a haustorium (the coconut sprout) inside the central cavity [18]. The haustorium absorbs the solid endosperm to nourish the seedling. Coconut fruits have two distinctive forms depending on the domestication. Wild coconuts feature has an elongated triangular fruit with a thicker husk and a smaller amount of endosperm. These allow the fruits to be more buoyant and make it easier for them to lodge into sandy shorelines, making their shape ideal for ocean. A full-sized coconut fruit weighs about 1.4 kilograms (3 pounds 1 ounce). Coconuts sold domestically in coconut-producing countries are typically not de-husked. Especially immature coconuts (6 to 8 months from flowering) are sold for coconut water and softer jelly-like coconut meat (known as “green coconuts”, “young coconuts”, or “water coconuts”), where the original coloration of the fruit is more aesthetically pleasing. [19]; [14]. The presence of PVP significantly alters the growth pattern of metallic magnesium structures that are produced when lithium naphthalenide reduces the Grignard reagents butyl and phenyl magnesium chloride. PVP causes the growth of well-separated faceted nanoparticles and large polycrystalline aggregates, respectively [20]. Also, a range of chemical and biological sensing platforms are made possible by the Held enhancement, which is also the foundation for improved spectroscopies like surface-enhanced Raman scattering (SERS [14]. Size, shape, and composition the materials all affect LSPRs. The noble metals Au and Ag have historically been the focus of plasmonic NP research, but more widely available, less expensive, and differently adjustable materials have surface as alternatives. As an illustration, magnesium is a desirable plasmonic metal because it can maintain resonances throughout the ultraviolet–visible–near–infrared (UV–vis–NIR) spectrum and because it is the eighth most plentiful element in the crust of the Earth [21]; [22].
Synthesis of metal oxide nanoparticles
Physical and chemical techniques have been primarily employed in the synthesis of metal oxide nanoparticles (MO NPs). Spray pyrolysis, ultrasonication, chemical vaporisation, and other physical techniques are examples [23]. Physical approaches, however, have numerous drawbacks, including inconsistent particle size and expensive gear [24]. Chemical synthesis of metal oxide nanoparticles can be achieved by precipitating the required metal ion precursor in an aqueous solution phase or by using an oxidation-reduction reaction [25]|. Conventional chemical procedures, although successful in producing a wide range of morphologies for inorganic nanoparticles, typically involve the use of hazardous chemicals that pose a risk to both human health and the environment. As a result, new, ecologically friendly synthetic techniques have been developed [26]. An extensive analysis of the biological synthesis of metal oxide nanoparticles employing various biomaterials is given in this paper. While chemical and physical approaches have traditionally dominated the synthesis of nanoparticles, a green chemistry-based approach to nanoparticle preparation is garnering significant attention particularly in this era of environmental and human health protection [27]. The production of green nanoparticles requires the utilization of biomolecules like as proteins, carbohydrates, and amino acids as well as microorganisms, plants, and living cells, as well as softer synthesis conditions such sol–gel processes [28].
Green synthesis
By enabling the synthesis of NPs at modest pressure, temperature, and pH as well as at a much reduced cost, green, or biological, NP synthesis eliminates many of the negative aspects [29]; [30]. It has been claimed that biomass filtrate from a variety of biological systems, including yeast, bacteria, actinomycetes, fungi, algae, and plant extract, can be used to create nanoparticles in a green manner. The production of distinct metal nanoparticles (NPs) of silicon, gold, zinc, titanium, copper, alginate, and magnesium has been studied in a variety of microorganisms, particularly bacteria and fungus [31]. According to a number of investigations, actinomycetes, bacteria, fungi, and viruses can biosynthesized metal nanoparticles (NPs) such as silver, gold, silver-gold alloy, tellurium, platinum, copper, zinc, selenium, palladium, silica, zirconium, quantum dots, titanium, and magnetite. The green synthesis of nanoparticles (NPs) is a bottom-up process in which biomolecules that are secreted, such as proteins, enzymes, polysaccharides, and carbohydrates, oxidize or reduce metallic ions to generate NPs. However, because different types of microorganisms interact with metallic ions in different ways, a comprehensive understanding of the process behind microbial NP production need to be achieved. The size, shape, and morphology of the synthesized NPs are ultimately influenced by the biochemical processing, the interaction activities of a particular microorganism, and the impact of environmental factors like pH and temperature. The aim of the study is to synthesize and characterize MgO nanoparticles using orange peel (citrus reticulata) and coconut shaft (Cocos Nucifera.L). Their biological activities are indicative of the optimization methods needed for the environmentally friendly synthesis of nanoparticles with particular sizes and shapes. Also, determining the role of each constituent in the biofabrication process demands full chemical analysis for biological biomass filtrate [32]. Further research is needed for commercial applications of scaled-up NP manufacturing. More explanations are needed on the mechanics of NP fabrication [33]; [34]. Conversely, in order to create novel commercial materials using green approaches to nanomaterial synthesis, basic science, chemical engineering, and industrial media must collaborate. Depending on the type of bacterium, either internal or extracellular processes can generate nanoparticles. Researchers have utilized living cell extracts for the biological production of nanoparticles. The subsequent sections provide a brief discussion of the primary biological pathways utilized in the synthesis of nanoparticles (NPs) [9].
2.0 Materials and Methods
2.1 Materials
Reagents/Materials: MgSO4, Distilled water, Sodium hydroxide pellets, Acetone, orange peels and coconut shaft.
2.2. Methodology
2.2.1. Determination of Moisture Content of the orange peel (citrus reticulata) and Coconut Mesocarp (shaft).
A 1.75 g of the orange peel (citrus reticulata) andcoconut shaft were weighed in different crucibles and heated for 2 h in a regulated oven at 105 oC to determine moisture present in the sample. The sample was for dried.
2.2.2. Preparation of orange peel (citrus reticulata) and Coconut Mesocarp Extract.
The orange peel (citrus reticulata) and coconut mesocarp were thoroughly washed with tap water, then distilled water, air dried for about 10 – 12 days. The shafts were pulverized into a fine particle using an electric blender then sieved off all impurities. 60 g of orange peel (citrus reticulata) and Coconut powder were weighed in a round bottom flask separately. 1.8 L of distilled water was added to it, add magnetic stirring bar into the solution, and subject it to boil using a magnetic stirrer hotplate for 2 h. 1 L, distilled water was added at intervals to prevent the solution from drying completely and help retain much extract. The solution was filtered using a filter paper to obtain the orange peel (citrus reticulata) and coconut mesocarp extract.
2.2.3. Preparation of the Metal Oxide Salt (MgO) Solution and Synthesis of MgO Nanoparticles
30 g of magnesium sulphate (MgSO4) was weighed into a 250 mL round-bottom flask and standardized with distilled water. Magnesiun oxide (MgO) solution was mixed with 600 mL of orange peel (citrus reticulata) and coconut mesocarp extract. After two h of boiling and continuous stirring in different round bottom flask, NaOH pellets was added, the mixture was monitored with a pH paper till a pH of 10 was obtained. The mixture was left to boil before being stirred for additional 2 h and then cooled, centrifuge for 15 min. at 400 rpm, to form a precipitate, washed repeatedly with acetone and distilled water to get rid of any impurities. Following a 24 hrs drying process, the gathered MgO particles were made into a powdery form and kept in an air tight nano bottle.
2.3 Characterization
X-ray diffraction (XRD) crystallinity and mineral phases of MgO NPs sample was identified using a Bruker D8 Advance X-ray diffractometer with Cu Kα radiation (45 kV, 40 mA, λ = 1.542 Å). Scan was conducted samples were scanned at 002s from the range of 10–80 (2θ). The surface morphology, composition and particle size of the MgO NPs was examined with both high resolution scanning electron microscopy-energy-dispersive X-ray spectroscopy (SEM-EDS) using a SEM HITACHI S-4700 electron microscope with spectral imaging system, The HRTEM is used to determine the internal morphology of the synthesized MgO NPs samples was conducted using Phillips Tecnai F20 super-twain. XRF elemental composition of MgO NPs was conducted using a Philips PW 2400 X-ray sequential spectrophotometer to determine their elemental constituents. The FTIR was used to determine the functional groups present in the MgO NPs sample with attenuated total reflectance (ATR). BET surface area of the MgO NPs was measured at a temperature of 77.35K using a quantachrome NOVA 2000 surface analyzer. Thermogravimetric analysis (TGA) was conducted to determine the thermal stability of the synthesized MgO NPs samples was conducted in air, at a heating rate of 10°C/min, by means of thermal analyses (TG-DTA) using a LINSEIS L81 apparatus.
3.0 Results and Discussion
3.1 Characterization
3.1.1 Thermogravimetric Analysis and FTIR Spectrometry
3.1.1 TGA and FT-IR Figure 1A presents the TGA thermal stability results of the synthesized nanoparticles of orange peel MgO NPs and coconut shaft MgO NPs with gradual loss in weight from 100 g sharp temperature decline at 333.72 oC and 308.29 oC respectively. The synthesized nanoparticles from orange peel were more stable than the coconut shaft. The FTIR results of functional groups identified in the synthesized MgO NPs from orange peel and coconut shaft in Fig. 1B revealed some common spectral absorption bands of selected functional groups at 3202.42–3209 cm-1, 2948.74–2895.69 cm -1, 2061.22–2058.77 cm-1, 1644.38–1628.85 cm-1, 1155.47–1002.65 cm-1, and 726.83–696.29 cm-1 corresponds to the stretching of these functional groups: O–H, C–H, C=O, C–O, Mg–O–Mg respectively while absorption bands between and 1435.03–1361.47 cm-1 corresponds to C–H bending.
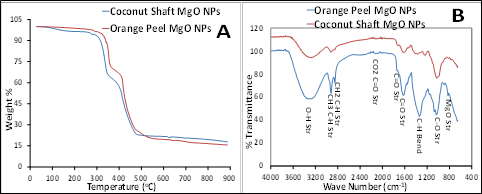
Figure 1: TGA analysis (A) and FTIR absorption bands (B) of synthesized MgO NPs from orange peels and coconut shaft extract
3.1.2 Scanning Electron Microscopy (SEM)
The SEM results of the synthesized MgO NPs from orange peels and coconut shaft in Figure 2 revealed spherical irregular particle shapes with agglomeration due to the presence of moisture. The coconut nanoparticles may likely have a low surface area to volume ratio which can be attributed to the clustering of the nanoparticle. The particle sizes of orange peel was about 50-60 nm while coconut shaft was 69-78 nm.

Figure 2: SEM images of synthesized MgO NPs from orange peel (A) and coconut shaft mesocarp (B) extract.
3.1.3 High Resolution Transmission Electron Microscopy
Fig. 3 presents the HRTEM of synthesized MgO NPs from orange peel and coconut shaft as it revealed spherical in shape with particle sizes of 3.42 nm and 4.84 nm respectively. The particle size of the orange MgO NPs was smaller than that of the coconut shaft which showed similar pattern with that of the SEM results and confirm irregular particle sizes with some agglomeration which can be attributed to the presence of moisture.
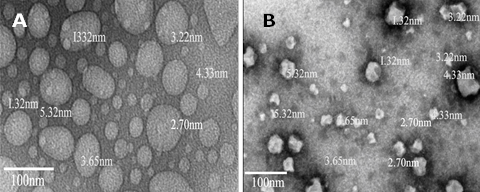
Figure 3: HRTEM images of synthesized MgO NPs from orange peel (A) and coconut shaft mesocarp (B) extract.
3.1.4 X-ray Diffraction (XRD)
X-ray diffraction (XRD) is a well-known technique for determining the crystal or amorphous structure of nanoparticles created using green synthesis approach such as coconut mesocarp extract. In the process of XRD analysis, a monochromatic beam of X-ray is indented on the sample and the diffraction pattern created by scattering of the rays is measured. XRD result in Fig. 4 revealed sharp peak spectrum for synthesized MgO NPs for orange peel and coconut shaft were highly polycrystalline nanoparticles which agrees with [35]; [36]; [37]; [38], The orange MgO NPs diffraction peaks at 2θ values of 34.3°, 37.99°, 44.08° and 64.69° are assigned to the (111), (220), (220), and (311) planes of a faced center cubic (FCC) lattice of magnesium oxide NPs while coconut NPs diffraction peak at 2θ values of 34.16°, 37.88°, 44.20° and 64.44° are assigned to the (010), (011), (002), and (022) planes of a faced center cubic (FCC) lattice of magnesium oxide NPs [39]. The composition of the mineral phases in orange peel MgO NPs consist of six minerals: Periclase (47 %), Quartz (25 %), Tenorite, Syn (2 %), Woodhouseite (3), Zincite (15 %), and Chlorargyrite (8 %). The composition of the mineral phases in coconut shaft (cocos nucifera.l) mesocarp MgO NPs consist of six minerals: Siderite Mg-be (24 %), Hanksite (28.8 %), Brucite (34.5 %), Chlorargyrite (8 %) quartz (4.6 %) and Woodhouseite (3 %). Periclase mineral phase was identified as MgO in the nanoparticles similar to the results of previous studies [40]; [41]; and brucite mineral phase identified as Mg(OH)2 [41]; [39]. The particle size of MgO NPs of the orange peel and coconut shaft was calculated using Debye Scherer’s formula for the particle size which were found to be 13 and 18 nm respectively [42].
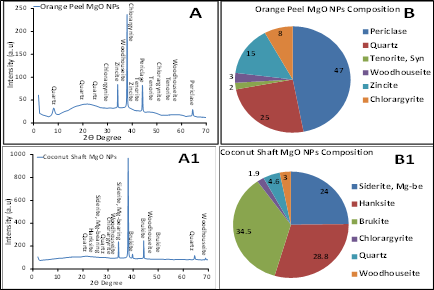
Figure 4: XRD spectra (A) and mineral composition (B) of synthesized MgO NPs from orange peel and coconut shaft extract
3.1.5 Brunauer-Emmet Teller (BET)
The BET results of synthesized MgO NPs from orange peels and coconut shafts are presented in Figures 4-8. The results revealed the BET parameters of synthesized orange MgO NPs are better than the coconut shaft MgO NPs. Fig. 5 A and B showed that the micropore and multi-point BET results of Orange peel was higher than the coconut shaft while Fig. 6A revealed the pore sized distribution of the MgO NPs and the cumulative surface area and pore volume of the orange peel was higher than that of coconut shaft while Fig. 6B revealed the multi-point BET of orange MgO NPs was higher than that of coconut shaft. The BET results of orange peel and coconut shaft MgO NPs are mesoporous and they all fall between the mesoporous range of BET analysis. The Langmuir showed steady increase for the orange peel while that of coconut increased and began to decline in Fig.7A while the surface area of orange peel was higher than coconut shaft in Fig. 7B. Fig. 8A revealed pore volume of thee orange peel was higher than the coconut shaft except for micropore while Fig. 8B revealed orange peel pore size was higher than that of coconut shaft except for pore diameter.
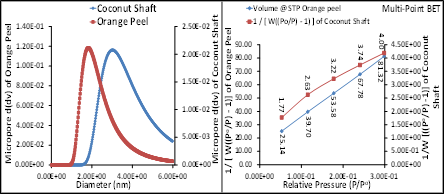
Figure 5: BET micropore (B) and multi-point BET (B) of synthesized MgO NPs from orange peel and coconut shaft extract

Figure 6: BET Pore size distribution (A) and DA method (B) of synthesized MgO NPs from orange peel and coconut shaft extract
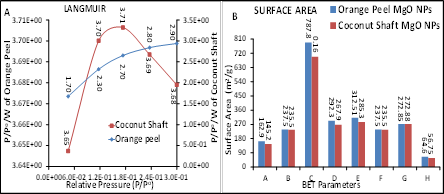
Figure 7: BET Langmuir (A) and BET Surface Area (B) of synthesized MgO NPs from orange peel and coconut shaft extract

Figure 8: BET Pore Volume (A) and Pore Size (B) of synthesized MgO NPs from orange peel and coconut shaft extract
3.1.6 XRF Elemental Composition
Fig. 9 is the XRF elemental composition of metal oxides present in the synthesized MgO NPs from orange peel and coconut shaft mesocarp. The result revealed MgO as the most dominant metal oxide at 45.52 % and 43.98 % for orange peel and coconut shaft magnesium oxide nanaoparticles respectively. Other constituents are silicon dioxide (SiO2), iron oxide (Fe2O3), aluminium oxide (Al2O3), and calcium oxide (CaO). The elemental composition of the other major metal oxides for orange peel synthesized MgO NPs were higher than the coconut shaft MgO NPs except for SiO2. The XRF elemental composition confirms Mg to be the dominant element as revealed in XRD mineral composition in Figure 4B.
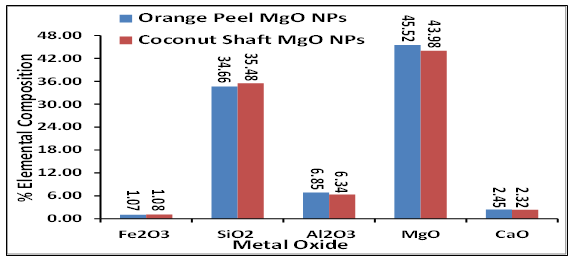
Figure 9: XRF elemental composition of synthesized MgO NPs from orange peel and coconut shaft extract
Conclusion
In conclusion, the biosynthesis of MgO NPs using orange peel (Citrus reticulata) and coconut mesocarp extract were successfully synthesized, and characterized as shown in the results as carried out in the study. The synthesis and characterization of MgO NPs were evaluated using various analytical techniques such as; FTIR which determined the functional groups present in the nanoparticles, SEM and HRTEM which revealed the spherical irregular particle morphology of the nanoparticles, BET which evaluated the surface area and pore size distribution of the nanoparticles which revealed that the orange peel was higher than coconut shaft, TGA of both nanoparticle samples were both thermally stable but hat of orange NPs was more stable than the coconut. XRD identified the crystallographic structure and mineral composition present in the nanoparticles. This analysis revealed that both the orange peel (Citrus reticulata) and coconut mesocarp extract operated as a low reducing agent and a good stabilizing agent, allowing for the generation of stable and biologically compatible magnesium oxide nanoparticles. XRF revealed Mg to the dominant element in the synthesized MgO NPs from orange peel and coconut shaft samples.
Acknowledgments
The authors acknowledged the contributions of LASU management for giving us the enabling environment to carry out the research, Mr. Ooluade, A (UNILAG) for the technical assistance.
Funding
This research work was carried out and financially supported by all the authors.
References
- Jeevanandam, J., Barhoum, A., Chan, Y. S., Dufresne, A., & Danquah, M. K. (2018). Review on nanoparticles and nanostructured materials: history, sources, toxicity and regulations. Beilstein journal of nanotechnology, 9(1), 1050-1074.
- Uskoković, V. (2023). Lessons from the history of inorganic nanoparticles for inhalable diagnostics and therapeutics. Advances in Colloid and Interface Science, 315, 102903.
- Ciriminna, R., Pagliaro, M., & Luque, R. (2021). Heterogeneous catalysis under flow for the 21st century fine chemical industry. Green Energy & Environment, 6(2), 161-166.
- Chadha, U., Selvaraj, S. K., Ashokan, H., Hariharan, S. P., Mathew Paul, V., Venkatarangan, V., & Paramasivam, V. (2022). Complex nanomaterials in catalysis for chemically significant applications: from synthesis and hydrocarbon processing to renewable energy applications. Advances in Materials Science and Engineering, 2022(1), 1552334.
- Hasan, S. (2015). A review on nanoparticles: Their synthesis and types. Research Journal of Recent Sciences, 4(ISC-2014), 1-3. www.isca.in, www.isca.me
- Wayman, T. M. R., Lomonosov, V., & Ringe, E. (2024). Capping agents enable well-dispersed and colloidally stable metallic magnesium nanoparticles. The Journal of Physical Chemistry. 128(11), 4666-4676. https://doi.org/10.1021/acs.jpcc.4c00366
- Talapin, D. V., & Shevchenko, E. V. (2016). Introduction: Nanoparticle chemistry. Chemical Reviews. 116(18), 10343–10345. https://doi.org/10.1021/acs.chemrev.6b00566
- Thakur, P., & Thakur, A. (2022). Introduction to nanotechnology. Synthesis and applications of nanoparticles, 1-17.
- Salem, S. S., & Fouda, A. (2021). Green synthesis of metallic nanoparticles and their prospective biotechnological applications: an overview. Biological trace element research, 199(1), 344-370.
- Chopra, H., Bibi, S., Singh, I., Hasan, M. M., Khan, M. S., Yousafi, Q., … & Cavalu, S. (2022). Green metallic nanoparticles: biosynthesis to applications. Frontiers in Bioengineering and Biotechnology, 10, 874742.
- Hornak, J. (2021). Magnesium oxide nanoparticles: Synthesis, properties, and selected technical applications. International Journal of Molecular Sciences, 22(23), 12752. https://doi.org/10.3390/ijms222312752
- Parveen, Z., & Mishra, S. (2021). Morphological and proximate analysis of orange (Citrus sinensis) peel and tomato (Solanum lycopersicum). European Journal of Nutrition & Food Safety. 13(5), 98-105. https://doi.org/10.9734/EJNFS/2021/v13i530423
- Armstrong, W. P. (2021). Edible Palm Fruits. In Wayne’s Word: An On-Line Textbook of Natural History. Palomar College. Retrieved April 20, 2021, from (https://web.archive.org/web/20180902172839/https://www2.polomar.edu/users/war mstrong/ecoph10.htm)
- Paull, R. E., & Ketsa, S. (2015, March). Coconut: Postharvest quality-maintenance guidelines (F_N-45). College of Tropical Agriculture and Human Resources, University of Hawai’i at Manoa. Retrieved from (https://www.ctahr.hawaii.edu/oc/freepubs/pdf/F_N-45.pdf)
- Niral, V., & Jerard, B. A. (2019). Botany, origin and genetic resources of coconut. In The Coconut Palm (Cocos nucifera L.)-Research and Development Perspectives (pp. 57-111). Singapore: Springer Singapore.
- Hasan, K. F., Horváth, P. G., Bak, M., & Alpár, T. (2021). A state-of-the-art review on coir fiber-reinforced biocomposites. Rsc Advances, 11(18), 10548-10571.
- Vieira, F., Santana, H. E., Jesus, M., Santos, J., Pires, P., Vaz-Velho, M., … & Ruzene, D. S. (2024). Coconut waste: discovering sustainable approaches to advance a circular economy. Sustainability, 16(7), 3066.
- Beveridge, F. C., Kalaipandian, S., Yang, C., & Adkins, S. W. (2022). Fruit biology of coconut (Cocos nucifera L.). Plants, 11(23), 3293.
- Nayar, N. M. (2017). In The coconut: Phylogeny, origins, and spread (pp. 10-21). Academic Press. ISBN 978-0-12-809778-6
- Vergheese, M., & Vishal, S. K. (2018). Green synthesis of magnesium oxide nanoparticles using Trigonella foenum-graecum leaf extract and its antibacterial activity. Journal of Pharmacognosy and Phytochemistry, 7(3), 1193-1200. https://doi.org/10.1016/j.jpp.2018.04.001
- Thomas, H. N. (2024). Metal tolerance in lichens. In Heavy Metal Tolerance in Plants (pp. 119-132). CRC Press.
- Duan, X., & Liu, N. (2019). Magnesium for dynamic nanoplasmonics. Accounts of chemical research, 52(7), 1979-1989
- Alhalili, Z. (2023). Metal oxides nanoparticles: general structural description, chemical, physical, and biological synthesis methods, role in pesticides and heavy metal removal through wastewater treatment. Molecules, 28(7), 3086.
- Shekunov, B. Y., Chattopadhyay, P., Tong, H. H., & Chow, A. H. (2007). Particle size analysis in pharmaceutics: principles, methods and applications. Pharmaceutical research, 24, 203-227.
- Hachem, K., Ansari, M. J., Saleh, R. O., Kzar, H. H., Al-Gazally, M. E., Altimari, U. S., … & Kianfar, E. (2022). Methods of chemical synthesis in the synthesis of nanomaterial and nanoparticles by the chemical deposition method: a review. BioNanoScience, 12(3), 1032-1057.
- Bhardwaj, B., Singh, P., Kumar, A., Kumar, S., & Budhwar, V. (2020). Eco-friendly greener synthesis of nanoparticles. Advanced pharmaceutical bulletin, 10(4), 566.
- Sati, A., Ranade, T. N., Mali, S. N., Ahmad Yasin, H. K., & Pratap, A. (2025). Silver nanoparticles (AgNPs): comprehensive insights into bio/synthesis, key influencing factors, multifaceted applications, and toxicity─ a 2024 update. ACS omega, 10(8), 7549-7582.
- Zikalala, N., Matshetshe, K., Parani, S., & Oluwafemi, O. S. (2018). Biosynthesis protocols for colloidal metal oxide nanoparticles. Nano-Structures & Nano-Objects. 16, 288–299. https://doi.org/10.1016/j.nanoso.2018.06.007.
- Ahmed, S. F., Mofijur, M., Rafa, N., Chowdhury, A. T., Chowdhury, S., Nahrin, M., … & Ong, H. C. (2022). Green approaches in synthesizing nanomaterials for environmental nanobioremediation: Technological advancements, applications, benefits and challenges. Environmental Research, 204, 111967.
- Dikshit, P. K., Kumar, J., Das, A. K., Sadhu, S., Sharma, S., Singh, S., … & Kim, B. S. (2021). Green synthesis of metallic nanoparticles: Applications and limitations. Catalysts, 11(8), 902.
- Abishagu, A., Kannan, P., Sivakumar, U., Manikanda Boopathi, N., & Senthilkumar, M. (2024). Metal nanoparticles and their toxicity impacts on microorganisms. Biologia, 79(9), 2843-2862.
- Kulkarni, D., Sherkar, R., Shirsathe, C., Sonwane, R., Varpe, N., Shelke, S., … & Dyawanapelly, S. (2023). Biofabrication of nanoparticles: sources, synthesis, and biomedical applications. Frontiers in Bioengineering and Biotechnology, 11, 1159193.
- Fernandes, C., Jathar, M., Sawant, B. K. S., & Warde, T. (2023). Scale-up of nanoparticle manufacturing process. In Pharmaceutical Process Engineering and Scale-up Principles (pp. 173-203). Cham: Springer Nature Switzerland.
- Shegokar, R., & Nakach, M. (2020). Large-scale manufacturing of nanoparticles—An industrial outlook. In Drug delivery aspects (pp. 57-77). Elsevier.
- Jhansi, K., Jayarambabu, N., Reddy, K. P., Reddy, N. M., Suvarna, R. P., Rao, K. V., … & Rajendar, V. (2017). Biosynthesis of MgO nanoparticles using mushroom extract: effect on peanut (Arachis hypogaea L.) seed germination. 3 Biotech, 7(4), 263. XA
- Safael, G. J., Zahedi, S., Javid, M., & Ghasemzadeh, M. A. (2015). MgO nanoparticles: an efficient, green and reusable catalyst for the onepot syntheses of 2, 6-dicyanoanilines and 1, 3-diarylpropyl malononitriles under different conditions. XH
- Ammulu, M. A., Viswanath, K. V., Giduturi, A. K., Vemuri, P. K., Mangamuri, U., & Poda, S. (2021). Phytoassisted synthesis of magnesium oxide nanoparticles from Pterocarpus marsupium rox. b heartwood extract and its biomedical applications. Journal of Genetic Engineering and Biotechnology, 19(1), 21. XI
- Fardood, S. T., Ramazani, A., & Joo, S. W. (2018). Eco-friendly synthesis of magnesium oxide nanoparticles using arabic Gum. J. Appl. Chem. Res, 12(1), 8-15. XY
- Taglieri, G., Daniele, V., & Mondelli, C. (2018). MgO nanoparticles synthesized starting from an innovative one‐step process. Journal of the American Ceramic Society, 101(4), 1780-1789.
- Agyapong, P. O., Gikunoo, E., Arthur, E. K., Anang, D. A., Agyemang, F. O., Foli, G., & Baah, D. S. (2023). Synthesis of magnesium oxide from waste magnesium-rich Cucurbita pepo (pumpkin) seeds. Sustainable Environment, 9(1), 2258473. XB
- Amin, M. A. A., Abu-Elsaoud, A. M., Nowwar, A. I., Abdelwahab, A. T., Awad, M. A., Hassan, S. E. D., … & Elkelish, A. (2024). Green synthesis of magnesium oxide nanoparticles using endophytic fungal strain to improve the growth, metabolic activities, yield traits, and phenolic compounds content of Nigella sativa L. Green Processing and Synthesis, 13(1), 20230215. XF
- Moorthy, S. K., Ashok, C. H., Rao, K. V., & Viswanathan, C. (2015). Synthesis and characterization of MgO nanoparticles by Neem leaves through green method. Materials Today: Proceedings, 2(9), 4360-4368. XC

Pingback: zyvox
Pingback: fluconazole over the counter canada
Pingback: lasix generic medication
Pingback: cialis mexico
Pingback: stendra discount
Pingback: metronidazole puppy reddit