Introduction: Fenamic acid-derived non-steroidal anti-inflammatory drugs (NSAIDs) are characterized by the N-phenyl anthranilic acid pharmacophore, which is further functionalized with substituents such as -Cl, -CF3, and -CH3, or their combinations, on one of the phenyl rings. These include well-known drugs like Fenamic acid, Flufenamic acid, Mefenamic acid, and Tolfenamic acid, as shown in Fig. 1. All these compounds belong to the class of aromatic amino acids and exhibit potent anti-inflammatory properties by targeting cyclooxygenase (COX) enzymes, particularly COX-1, to inhibit prostaglandin synthesis. However, the non-selective inhibition of COX-1 by these NSAIDs is associated with the adverse side effect of peptic ulcer formation. To mitigate these ulcerogenic effects, research has focused on modifying the structure of fenamic acid derivatives. One approach has been to mask the free carboxyl group (-COOH) by developing ester prodrugs or creating amide, ester, and ether derivatives [1]. These modifications aim to improve the pharmacokinetic and pharmacodynamic profiles of these drugs, providing better therapeutic outcomes with reduced gastrointestinal toxicity.
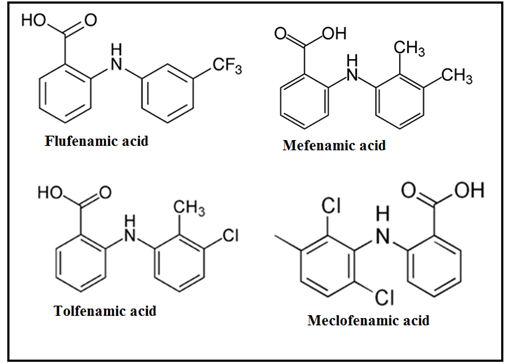
Fig. 1 Structure of Fenamates: Flufenamic, Mefenamic, Tolfenamic and Meclofenamic acid
Fenamic acid-derived NSAIDs, including analogues such as Tolfenamic acid, Mefenamic acid, and Flufenamic acid, have emerged as promising candidates for a range of therapeutic applications beyond their well-established role in inflammation management. Recent studies have identified these compounds as potent neuroprotective, immune-modulatory, and anticancer [2] agents, thanks to their ability to target multiple, synchronized regulatory pathways involved in disease pathogenesis. In addition to their traditional role in targeting cyclooxygenase enzymes, fenamates have been repurposed for the management of neurodegenerative diseases such as Alzheimer’s, Huntington’s, epilepsy, and stroke [3]. Their therapeutic action extends beyond direct inhibition of ion channels and enzymes involved in these conditions to include modulation of transcriptional factors that influence gene expression related to disease processes.
Further investigations into the mechanism of action of fenamates have revealed their potential to suppress NLRP3 inflammasome activation and promote proteasome-dependent SP1 degradation, indicating that their pharmacophore [4] is not restricted to NSAID activity alone. Chemically modified fenamate analogues also exhibit activity against a range of other molecular targets, including AKR1C2, AKR1C3, topoisomerase-1, TRESK channels, and have shown promise in anti-HIV and anti-leukemic applications.
In the context of drug repurposing, fenamic acid-derived NSAIDs present a robust and attractive option due to their exceptional biological profiles in both native and modified forms. Drug repurposing offers an economically sustainable approach to drug development by reducing the need for extensive de novo synthesis efforts. Moreover, these NSAIDs, particularly Tolfenamic acid, Mefenamic acid, and Flufenamic acid, have been identified as potential candidates in the fight against multi-drug resistant microbes an area of critical importance, given the WHO’s warning that antimicrobial resistance poses a significant global health threat. Despite these promising preclinical findings, the clinical application of chemically modified fenamic acid-derived NSAIDs remains largely unexplored, with few candidates in the clinical pipeline. This highlights the need for further investigation into the full potential of these compounds, which could lead to novel therapies for a broad spectrum of diseases [5-12].
Recent developments in the synthesis and characterization of metal complexes of mefenamic acid and other fenamates have highlighted their promising potential for a variety of applications in medicinal chemistry and beyond. Notably, the complex of mefenamic acid with bismuth (III) [13] exhibits excellent in vitro activity against Helicobacter pylori, the bacterium implicated in the development of gastric, peptic, and duodenal ulcers, as well as gastric cancer. This finding underscores the potential of metal-based fenamate complexes in combating gastrointestinal diseases [14].
Furthermore, mefenamic acid complexes with metal ions such as Mn(II), Co(II), Ni(II), Zn(II), and Cu(II) have shown significant antioxidant properties and inhibition of soybean lipoxygenase [15], an enzyme involved in inflammation. Among these, the Mn(II) complex stands out for its remarkable antioxidant and anti-inflammatory effects, while the Cu(II) complex demonstrates antitumor activity comparable to the widely used chemotherapeutic agent cisplatin. These results open new avenues for the use of mefenamic acid-metal complexes in cancer therapy and inflammation management.
In addition, Feng et al. [16] reported that mefenamic acid complexes with Mn(II) exhibit enhanced inhibitory activity against lipoxygenase compared to the parent ligand, mefenamic acid, further suggesting the efficacy of these metal derivatives in inflammatory conditions. Additionally, complexes of mefenamic acid with Ni(II), Zn(II), and Cu(II) show high affinity for bovine and human serum albumins, which could be advantageous for drug delivery applications.
Most of these complexes are mononuclear, synthesized in varying metal-to-ligand (M: L) ratios, including 1:1, 1:2, 1:3, and 1:4. Interestingly, studies on mefenamic acid complexes with Fe(III) ions have revealed the formation of both mononuclear and binuclear complexes[17], depending on the metal-to-ligand ratio, further expanding the scope of fenamate-metal interactions and their possible biological implications.
Copper(II) complexes of non-steroidal anti-inflammatory drugs (NSAIDs) have emerged as attractive alternatives to other metal-based systems owing to the low cost, redox activity, and biological relevance of copper salts. Compared to the free ligands, copper (II) NSAID complexes often exhibit enhanced biological performance [18-25], including improved antioxidant, anti-inflammatory, and anticancer activities. Among NSAIDs, mefenamic acid and other fenamates are particularly versatile ligands due to the presence of a carboxylic acid moiety [26-32], which enables multiple coordination modes depending on the metal-to-ligand ratio, choice of co-ligand, and reaction conditions such as solvent, temperature, and pH.
Beyond their biological importance, carboxylate-containing ligands are also of considerable interest in coordination chemistry and crystal engineering, as they readily form strong hydrogen-bonding interactions with nitrogen-donor heterocycles. Such interactions facilitate the construction of supramolecular architectures and hydrogen-bonded networks [33-41]. The growing use of nitrogen-donor co-ligands in metal complexes has further expanded the structural diversity of carboxylate-based systems and significantly influenced their coordination behavior, redox properties, and biochemical activity.
Despite extensive biological studies on fenamate metal complexes, systematic electrochemical investigations correlating ligand-based redox behavior with copper (II) coordination remain limited. In the present work, a comprehensive cyclic voltammetric and spectroscopic study of fenamic acid and its derivatives flufenamic acid, mefenamic acid, meclofenamic acid, and tolfenamic acid along with their copper(II) complexes in a 1:2 (metal:ligand) ratio is reported for the first time in a non-aqueous medium (DMSO) using a glassy carbon electrode. The electrochemical data reveal distinct interactions between copper (II) ions [42] and fenamate ligands, manifested by systematic shifts in redox potentials upon complexation.
This study provides new insights into ligand-centered [42] versus metal-assisted redox processes in copper (II) fenamate systems, thereby establishing a direct link between coordination chemistry and electrochemical behavior. The findings highlight the electroactive nature of fenamate-based copper complexes and underline their potential relevance in redox-mediated biological and catalytic applications.
MATERIAL AND METHODS
Reagent and solution
A key novelty of the present investigation lies in the use of high-purity, analytically certified fenamic acid derivatives and copper (II) salts to ensure reproducible and interference-free electrochemical and spectral measurements. All fenamate ligands fenamic acid, flufenamic acid, mefenamic acid, clofenamic acid, and tolfenamic acid were sourced as analytical or HPLC-grade standards and employed without further purification, thereby eliminating ambiguities arising from impurities or degradation products that often complicate electrochemical interpretations. The exclusive use of HPLC-grade dimethyl sulfoxide (DMSO) as a non-aqueous medium provides a stable electrochemical window and enables a direct comparison of ligand-centered and metal-assisted redox processes across structurally related fenamate systems. This controlled experimental design allows, for the first time, a systematic and comparative evaluation of the intrinsic electrochemical behavior of fenamate ligands and their copper (II) complexes, establishing reliable structure–redox correlations that are rarely addressed with such rigor in fenamate-based coordination chemistry. Stock solutions of each no steroidal anti-inflammatory drug (NSAID) were prepared at a concentration of 2 mM by accurately weighing the appropriate amount of each compound and dissolving it in HPLC-grade DMSO using volumetric flasks.
Apparatus
The cyclic voltammetric studies of fenamates ligand and their complexes were done by the BAS electrochemical three electrodes systems, EPSILON model. The procedure of electrochemical methods was published elsewhere [43]. The software driven PerkinElmer electronic absorption spectroscopy model LAMBDA-35 was employed for all the electronic absorption spectral studies. This is scanning double –beam spectrophotometer for UV/Vis range; operation by PC. The stock solution (1.0 × 10⁻² mol. L-1) of all free ligands and its copper (II) complexes were prepared in the DMSO non aqueous solvent. More dilute (1 x 10-3 mol. L-1) solutions were prepared by accurate dilution. Perhaps the most commonly used method [44] to study the interaction of free ligands with metal is the electronic absorption spectroscopy.This technique is based on monitoring changes occur in the spectrum of ligands or its metal complexes. The methodology [45] and devices of Mass spectroscopy (MS) Q-P50-A-D-I analysis Shimadzu QP (E170Eν)-2010-Pluss spectrometer has been used to collect mass spectra for fenamates and their complexes with copper (II) metal ion.
Preparation of complexes solutions
All solutions and dispersions of the various fenamate compounds were prepared using DMSO as the solvent. Stock solutions of the fenamate ligands—fenamic acid (FA), clofenamic acid (CFA), flufenamic acid (FFA), mefenamic acid (MFA), and tolfenamic acid (TFA) were prepared in DMSO at concentrations ranging from 250 μ mol. L-1 to 1.0 mol. L-1, as required. Standard stock solutions (1.0 × 10⁻² mol. L-1) of each ligand and their corresponding metal complexes were prepared in DMSO, using a 1:2 molar ratio of metal to ligand, respectively. More dilute solutions (1.0 × 10⁻³ mol. L-1) were prepared by accurate dilution of the stock solutions for use in cyclic voltammetry, mass spectrometry, and electronic absorption spectroscopy.
RESULT AND DISCUSSION
Spectroscopic evidence of coordination-driven electronic modulation in fenamate–copper (II) systems in a non-aqueous medium
The electronic absorption spectral data of various non-steroidal anti-inflammatory drugs (NSAIDs) and their copper (II) complexes reveal interesting insights into their electronic transitions in dimethylsulfoxide (DMSO) at 25°C. The fenamate ligands fenamic, mefenamic, clofenamic, flufenamic, and tolfenamic acids show distinct electronic absorption bands ranging from 282 nm to 703 nm.
The electronic absorption spectral data for various NSAIDs and their corresponding copper (II) complexes, prepared in a 1:2 metal-to-ligand molar ratio in dimethylsulfoxide (DMSO), are presented in Table 1.
Low-Energy Transitions:
- The bands at 282-287 nm are attributed to π–π* transitions [40] in the pyridine ring of the NSAIDs.
- The 345-356 nm band corresponds to n–π* transitions associated with the -COOH group, which is present in all fenamates structures.
d–d Transitions:
The low-intensity bands observed at 671 nm to 703 nm for the NSAIDs are ascribed to d–d transitions [40] in the copper (II) complexes. These bands confirm the coordination of the metal with the ligands [40].
Copper (II) Complexes:
- The copper (II) complexes exhibit additional high-energy transitions around 457-461 nm, likely corresponding to metal-to-ligand charge transfer (MLCT) transitions.
- Given that six-coordinate copper complexes typically show absorption bands within the range of 671 nm to 703 nm, it supports the conclusion that the copper (II) complexes with fenamate ligands exhibit an octahedral geometry [40] in DMSO.
This analysis indicates a well-defined interaction between the NSAIDs ligand and copper (II), contributing to our understanding of the electronic properties and structural characteristics of these complexes.
Table 1: Comparison of ligand-centered and metal-assisted electronic transitions in fenamates Cu (II) systems (1:2) recorded in DMSO
| Compound | Color of compound | Ratio | λmax | Assignment |
| Fenamic Acid (FA) | Color less | – | 286, 353 | π- π*, , n-π* |
| Mefenamic Acid (MFA) | Color less | – | 282, 356 | π- π*, n-π* |
| MeClofenamic Acid (MCFA) | Color less | – | 287, 345 | π- π*, n-π* |
| Fluefenamic Acid (FFA) | Color less | – | 284, 345 | π- π*, n-π* |
| Tolfenamic Acid (TF) | Color less | – | 241, 286, 350 | metal–ligand electronic coupling |
| Cu(FA)2 | Pale greenish-yellow | 1: 2 | 681, 461 | d – d, Ligand-to-Metal Charge Transfer |
| Cu(MFA)2 | Pale greenish-yellow | 1: 2 | 671, 457 | d – d, Ligand-to-Metal Charge Transfer |
| Cu(MCFA)2 | Pale greenish-yellow | 1: 2 | 674, 457 | d – d, Ligand-to-Metal Charge Transfer |
| Cu(FFA)2 | Pale greenish-yellow | 1: 2 | 681, 461 | d – d, Ligand-to-Metal Charge Transfer |
| Cu(TA)2 | Pale greenish-yellow | 1: 2 | 703, 457 | d – d, Ligand-to-Metal Charge Transfer |
Mass spectral studies of fenamates ligand and copper(II) complexes
Under the mass spectral study it gives a final peaks in the ligand, 213.2, 241, 296.1, 281.1 and 261, respectively and other peaks at 208, 209.08, 223 229.02, 244.05 and 245 amu (Fig.2) may be due to different fragment. The intensity of these peaks gives an idea of the stability of these fragments [43-48].
The peaks in copper (II) complexes with fenamate and its derivatives give a final peak at 597, 634.5, 726.5, 733, and 692.5 amu and other peaks at 208, 209.02, 223, 229. 09, 244.05, 261 amu (Fig.2) may be attributed to different (ligand) fragment, which support the coordination of ligand to the metal ion and the structure of copper (II) complxes.
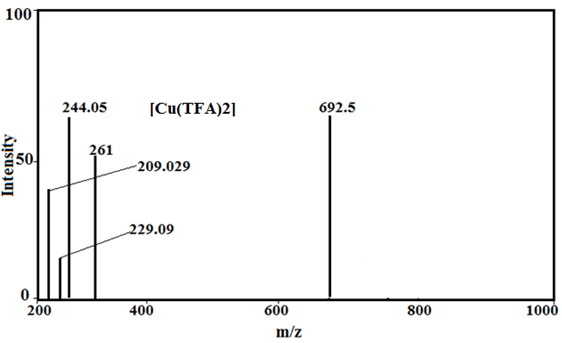
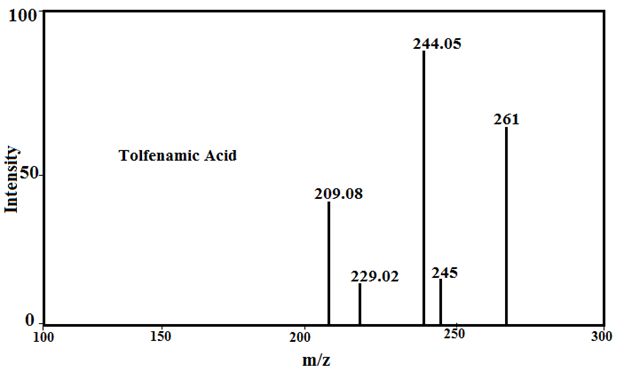
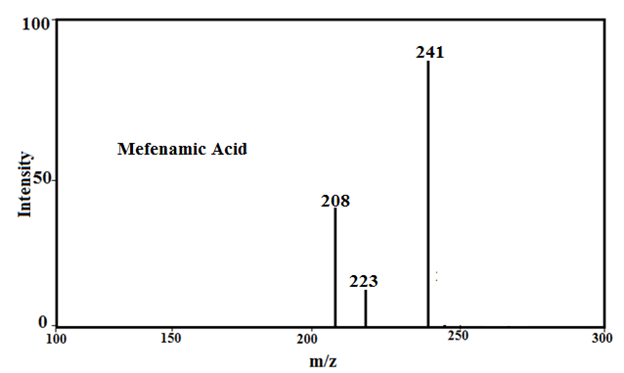
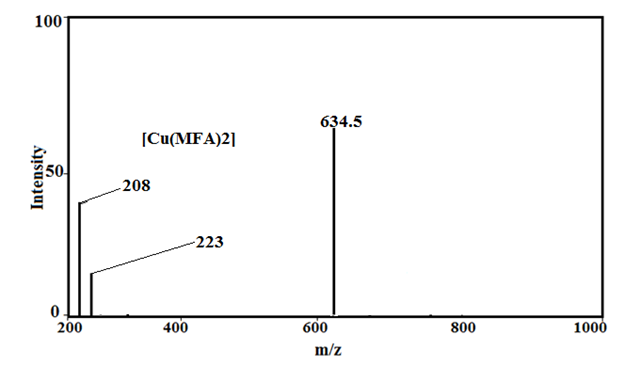
Fig. 2: Mass spectra of Tolfenamic acid, Mefenamic acid, [Cu(TFA)2] and [Cu(MFA)2]
Cyclic voltammetry
(i) Cyclic voltammetric behaviour of Fenamates (NSAIDs) ligand in non aqueous medium
The redox behavior of fenamates (NSAIDs) has been investigated using cyclic voltammetry (CV) in a non-aqueous medium under nitrogen conditions, with a potential range from 0.0 mV to -1800 mV versus Ag/AgCl reference electrode. The findings, detailed in Table 2 and Fig. 3, reveal the following characteristics: Fenamic Acid: Exhibits two irreversible reduction waves:Epc1 = -1040 mVand Epc2 = -1508 mV; Mefenamic Acid: Shows two reduction waves:Epc1 = -860 mV and Epc2 = -1330 mV;
Meclofenamic and Flufenamic Acids
Meclofenamic acid (MCFA) exhibits two overlapping cathodic reduction waves at Epc1 = –785 mV and Epc2 = –1140 mV. Similarly, flufenamic acid displays two distinct reduction peaks at Epc1 = –1202 mV and Epc2 = –1288 mV. During the anodic scan from –1800 mV to +1000 mV, both compounds show a single, ill-defined irreversible anodic peak (Epa) at 433 mV (MCFA) and 263 mV (flufenamic acid), respectively. In contrast, tolfenamic acid presents only a single reduction wave at Epc = –1253 mV (see Table 3).
It is important to note that an anodic scan initiated from 0.0 mV to +1000 mV did not produce any anodic peak potential (Epa) for the fenamate (NSAID) ligands. These ligands exhibited only reduction processes, characterized by cathodic peaks (Epc1 and Epc2), consistent with an EC (electrochemical-chemical) mechanism[40–43].
Table 2: CV data for 5 mM FA, MFA, MCFA, FFA and TA in dimethylsulfoxide containing 0.2 M NaClO4, (FAs= Fenamic acids)
| Scan rate (mVs-1) | Epc1 (mV) | Ipc1 (µA) | Epc2 (mV) | Ipc2 (µA) | Epa (mV) | Ipa (µA) |
| Fenamic Acid (FA) | ||||||
| 25 | -1040 | 12.3 | -1508 | 7.0 | 367 | 3.8 |
| 50 | -1047 | 17.5 | -1525 | 10.2 | 379 | 4.4 |
| 100 | -1059 | 25.0 | -1538 | 13.8 | 410 | 7.1 |
| 200 | -1069 | 25.8 | -1558 | 14.2 | 440 | 7.8 |
| Mefenamic Acid (MA) | ||||||
| 25 | -860 | 8.6 | -1330 | 5.4 | 730 | 3.0 |
| 50 | -868 | 12.4 | -1363 | 6.3 | 744 | 5.8 |
| 100 | -885 | 17.2 | -1388 | 9.0 | 749 | 8.0 |
| 200 | -898 | 17.8 | -1400 | 9.8 | 754 | 9.0 |
| Meclofenamic Acid (MCFA) | ||||||
| 25 | -785 | 1.4 | -1140 | 11.0 | 433 | 5.0 |
| 50 | -835 | 1.8 | -1162 | 16.4 | 438 | 5.8 |
| 100 | -867 | 2.3 | -1172 | 17.8 | 468 | 6.8 |
| 200 | -870 | 2.8 | -1178 | 20.7 | 478 | 9.2 |
| Fluefenamic Acid (FFA) | ||||||
| 25 | -1202 | 8.8 | -1288 | 13.7 | 263 | 4.8 |
| 50 | -1210 | 9.7 | -1292 | 18.5 | 263 | 5.7 |
| 100 | -1217 | 10.2 | -1298 | 25.7 | 267 | 6.4 |
| 200 | -1220 | 10.8 | -1307 | 30.2 | 270 | 8.3 |
| Tolfenamic Acid (TA) | ||||||
| 25 | -1253 | 13.4 | – | – | 496 | 3.2 |
| 50 | -1296 | 18.2 | – | – | 504 | 4.3 |
| 100 | -1298 | 26.3 | – | – | 508 | 7.0 |
| 200 | -1303 | 32.1 | – | – | 510 | 7.5 |
The NSAID ligands exhibit redox activity primarily at the aromatic rings. It is proposed that the single anodic peak (a) observed may result from the oxidation of electrochemically generated species formed during the reduction processes at Epc1 or Epc2, consistent with an EC (electrochemical–chemical) mechanism \[19–22, 47–52].
Based on the first reduction potential (Epc1) values of the fenamate (NSAID) ligands (Table 3), the ease of reduction increases in the following order: TFA → FFA (ill-defined) → FA → MFA → CFA (ill-defined).
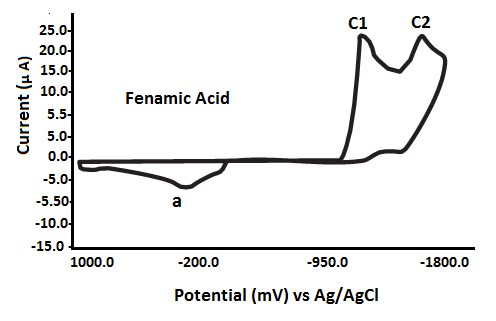
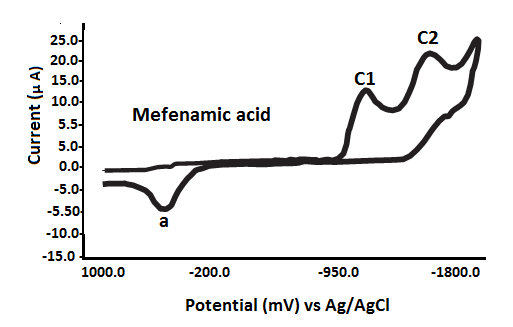
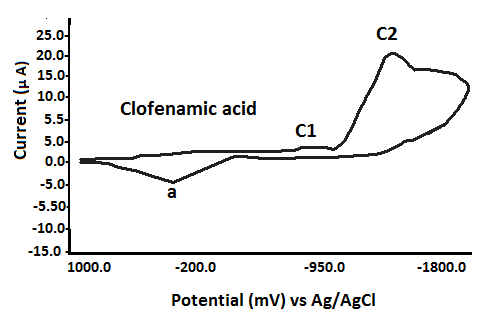
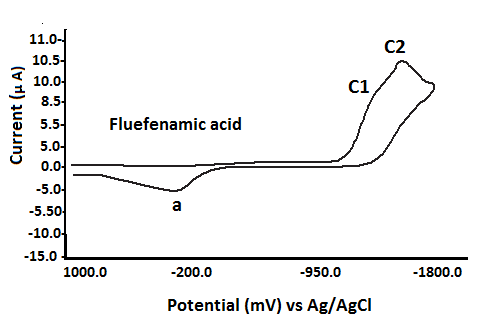
Fig. 3 Cyclic voltammograms of various fenamate (Fenamic, Mefenamic, MeClofenamic and Flufenamic acid) ligands in DMSO with 0.2 M NaClO4 supporting electrolyte at GCE versus Ag/AgCl reference electrode
(ii) Cyclic voltammetric behavior of Cu (II)–fenamate complexes (1:2 metal–ligand ratios) in DMSO
The cyclic voltammetric data for copper (II) complexes with fenamate (NSAID) ligands are presented in Table 3 and Figure 4. The Cu(II) complexes with fenamic acid (FA) and mefenamic acid (MFA) exhibit similar electrochemical behavior, showing two irreversible cathodic peaks at Epc1 = –875 mV and Epc2 = –1310 mV during the forward scan (0.0 mV to –1500 mV). In the reverse scan (–1500 mV to +1000 mV), a single irreversible anodic peak is observed at Epa1 = –625 mV for the FA complex and –580 mV for the MFA complex, recorded at a scan rate of 25 mV s⁻¹ (see Table 4).
In contrast, the Cu (II) complexes with clofenamic acid (CFA) and flufenamic acid (FFA) display only one cathodic peak (Epc1) and one anodic peak (Epa1), indicating simpler redox behavior. The Cu (II) complex with tolfenamic acid (TFA) exhibits two cathodic waves at Epc1 = –945 mV and Epc2 = –1198 mV. These electrochemical responses are attributed to ligand-centered redox processes. This assignment is supported by the similarity in voltammetric profiles between the free fenamate ligands and their corresponding copper (II) complexes [21, 22, 47].
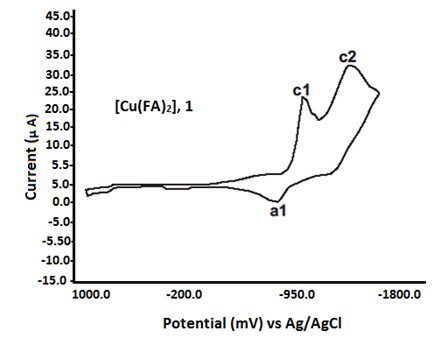

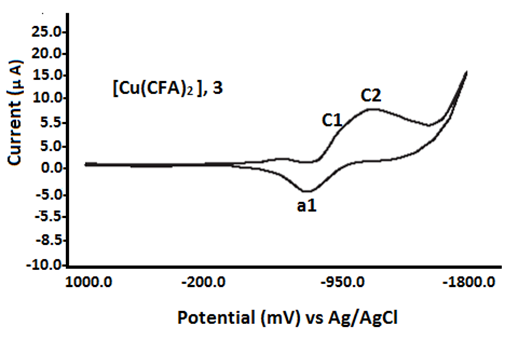

Fig. 4 Redox characteristics of Cu (II) fenamate complexes (1:2)
Table 3: CV data for copper (II) complexes with FA, MFA, MCFA, FFA and TFA in 1: 2 molar ratios in dimethylsulfoxide (DMSO) containing 0.2 M NaClO4
| Scan rate (mVs-1) | Epc1 (mV) | Epa1 (mV) | Ipc1 (µA) | Ipa (µA) | E0’ (mV) | ΔEp (mV) | Epc2 (mV) | Ipc2 (µA) |
| Cu(FA)2 | ||||||||
| 25 | -875 | -625 | 11.6 | 2.8 | -750.0 | 250 | -1180 | 7.7 |
| 50 | -900 | -617 | 15.2 | 3.5 | -758.5 | 283 | -1200 | 12.8 |
| 100 | -920 | -608 | 24.2 | 4.7 | -764.0 | 312 | -1220 | 18.7 |
| 200 | -930 | -600 | 34.1 | 5.5 | -765.0 | 330 | -1234 | 20.4 |
| Cu(MFA)2 | ||||||||
| 25 | -950 | -580 | 8.0 | 2.8 | -765.0 | 370 | -1310 | 1.5 |
| 50 | -975 | -590 | 10.0 | 3.7 | -782.5 | 385 | -1332 | 1.8 |
| 100 | -1037 | -632 | 15.5 | 5.8 | -834.5 | 400 | -1354 | 3.0 |
| 200 | -1087 | -524 | 17.5 | 6.8 | -804.0 | 563 | -1380 | 3.8 |
| Cu(CFA)2 | ||||||||
| 25 | -655 | -478 | 7.5 | 2.8 | -566.5 | 177 | – | – |
| 50 | -702 | -475 | 13.8 | 3.9 | -588.5 | 227 | – | – |
| 100 | -728 | -473 | 15.7 | 6.0 | -600.5 | 245 | – | – |
| 200 | -748 | -470 | 18.0 | 6.8 | -609.0 | 278 | – | – |
| Cu(FFA)2 | ||||||||
| 25 | -1003 | -698 | 5.5 | 2.5 | -850.5 | 305 | – | – |
| 50 | -1015 | -695 | 8.0 | 3.6 | -855.0 | 380 | – | – |
| 100 | -1040 | -692 | 11.5 | 4.2 | -866.0 | 384 | – | – |
| 200 | -1062 | -600 | 12.5 | 5.2 | -831.0 | 462 | – | – |
| Cu(TFA)2 | ||||||||
| 25 | -945 | -830 | 20.2 | 9.3 | -887.5 | 115 | -1198 | 3.2 |
| 50 | -1010 | -830 | 20.8 | 11.2 | -920.0 | 180 | -1198 | 3.8 |
| 100 | -1105 | -828 | 21.1 | 14.9 | -966.5 | 277 | -1198 | 4.8 |
| 200 | -1155 | -825 | 21.9 | 17.8 | -990.0 | 330 | -1200 | 5.2 |
Proposed structure of fenamates copper(II) complexes
The copper (II) complexes with various fenamate (NSAID) ligands were prepared in a 1:2 metal-to-ligand molar ratio in a non-aqueous medium and characterized using UV-Visible spectroscopy and electrochemical methods. Spectral analysis indicates that the coordination of the fenamate ligands to the copper(II) ion occurs in a bidentate manner. The data suggest that these complexes adopt a distorted octahedral geometry around the copper (II) center in DMSO, which is likely influenced by the coordination of solvent molecules (Figure 5).
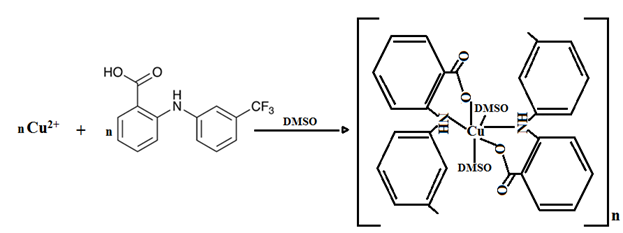
Fig. 5 Proposed structure of fenamates copper(II) complexes ( fenamate = fluefenamic acid)
Conclusion
Cyclic voltammetry and spectral studies play a crucial role in exploring the redox behavior, stability, and coordination characteristics of metal complexes derived from fenamic acid and its analogues, especially in non-aqueous media. These techniques provide key insights into electron transfer mechanisms, ligand field interactions, and coordination dynamics factors that are fundamental to the design of novel anti-inflammatory, analgesic, and anticancer agents. Moreover, such investigations enhance our understanding of the electrochemical properties of these pharmaceutically relevant compounds.
This study presents a comprehensive electrochemical and spectroscopic investigation of fenamic acid and its derivatives coordinated to copper(II) ions in a 1:2 metal-to-ligand molar ratio, conducted in a non-aqueous medium (DMSO) using a glassy carbon electrode (GCE). The copper(II) fenamate complexes were characterized by UV–Visible and mass spectroscopy, confirming bidentate coordination of fenamate ligands and revealing a distorted octahedral geometry around the copper center, influenced by coordinated solvent molecules.
Electrochemical analysis showed that free fenamic acids exhibit irreversible cathodic peaks and a single anodic peak, while their copper (II) complexes display ligand-centered redox behavior, evidenced by striking similarities in cyclic voltammograms between the free ligands and complexes. This highlights the dominant role of the fenamate ligands in the electrochemical response.
Importantly, the cyclic voltammetry method using GCE proved to be a simple, cost-effective, and reliable analytical tool for quantitative determination of these NSAIDs, with potential applications in pharmaceutical analysis.
The stability of the copper(II) fenamate complexes was found to depend on multiple factors, including the oxidation state of the metal center, ligand nature and steric effects, chelation strength, as well as external conditions such as temperature, pH, and solvent environment. Overall, this work provides valuable insights into the coordination chemistry and electrochemical behavior of copper (II) fenamate complexes, establishing a foundation for their future applications in drug analysis and bioinorganic chemistry.
Acknowledgment
I (A.K.S.) gratefully acknowledge Prof. (Dr.) Krishna Srivastava (Former Head) and Prof. (Dr.) Jagdish Prasad (Former Professor), Department of Chemistry, University of Allahabad, for their affection, encouragement, and keen interest in my research work. I also extend my sincere thanks to Prof. (Dr.) Ashutosh Srivastava, Director, BBSCET, Prayagraj, for his constant support and for providing a conductive environment for carrying out this research.
Funding There is no fund to mention for this research.
Conflict of interest there is no conflict of interest to declare.
References
- A. Sharma, M., Prasher, P., Synthesis and medicinal applications of fenamic acid derivatives, Current Organic Chemistry. 27, 1132-1142 (2023)
- Oshite , Y., A. Hirai, W., Ichii, R., Kuroda, C., Hasegawa, K., Hiroshige, R., Yokoyama, H., Tsuchida, T., Goto, S., Comparative study on the effects of the inclusion complexes of non-steroidal anti-inflammatory drugs with 2-hydroxypropyl-β-cyclodextrins on dissociation rates and supersaturation. RSC Pharm. 1, 80-97 (2024)
- LuísP.G. Monteiro, A. Gomes, C. Silva, F. Martel, Santos,T.M. , Ferreira,B.J.M.L. Towards targeted cancer therapy: Synthesis, characterization, and biological activity of a new Cu (II)-ibuprofen-2,2′-dipyridylamine metal complex. Heliyon, 11 (1), 1-11 (2025)
- González, F .D. Madrid, F. S., NSAIDs: Learning new tricks from old drugs. Eur. J. Immunol. 45(3), 679-686 (2015)
- Lazou, M. , Hatzidimitriou, A. G. , A. Papadopoulos, N. , Psomas, G. , Transition metal(II) complexes with the non steroidal anti–inflammatory drug oxaprozin: Characterization and biological profile. J. of Inorg Biochem, 243, 112196 (2023)
- Malis, G., A. Bakali, S. , Hatzidimitriou, A. G. , Psomas, G. , Copper(II) complexes with non-steroidal anti-inflammatory drugs and neocuproine: Structure and biological evaluation, Journal of Molecular Structure. 1303, 137590 (2024)
- Nnabuike, G. G., Gawali, S. S.- , Patil, A. S. , R. Butcher, J. , Obaleye, J. A., Ashtekar, H. , Prakash, B., Copper(II) complexes containing derivative of aminobenzoic acid and nitrogen-rich ligands: Synthesis, characterization and cytotoxic potential, Journal of Molecular Structure. 1279, 135002 ( 2023)
- Valentovic M., “Mefenamic Acid Monica.” Pharm: The Comprehensive Pharmacology Reference (Elsevier, New York, NY) 1 (2007)
- Shetti, N. P., Nayak, D. S., Malode, S. J., R. Kulkarni, M., Kulkarn,i D. B., Teggi, R. A., Joshi, V. V., Surfaces and Interfaces. 9, 107 (2017)
- Shetti, N. P., Nayak, D. S., Malode, S. J., Kulkarni, R. M., J. Electrochem. Soc.164(5) B3036-B3042, (2017)
- Shetti, N. P., Nayak, D. S., Malode, S. J., K. Reddy, R., Shukl,a S. S., Aminabhavi, T. M., Colloids Surfaces B Biointerfaces. 177, 407 (2019)
- Ahmed, S., Sheraz, M. A., Ahmad, I., “Tolfenamic Acid.” Profiles of Drug Substances, Excipients and Related Methodology (Elsevier, Oxford, London). 43, 255 (2018)
- Daniels, M. J. D. et al., Nat. Commun.7, 24 (2016)
- Lago, E. M. et al., E Bio Medicine. 43, 370 (2019)
- Seong, Y. J., Alhashim, M., A. Mayhoub, Mohammad, H., Seleem, M. N., Antimicrob. Agents Chemother. 64, 1 (2020)
- Ozkan, S. A., Uslu, B., J. Pharm. Biomed. Anal. 130, 126 (2016)
- Abo El-Maali, N., Bioelectrochemistry. 64, 99 (2004)
- Hasanzadeh, M., Shadjou, TrAC – Trends, N., Anal. Chem. 80, 167 (2016)
- Baranowska, I., Markowsk,i P., Gerle, A., Baranowski, J., , Bioelectrochemistry. 73, 5 (2008)
- Amor-García, I., Blanco-López, M. C., Lobo-Castañón, M. J., Miranda-Ordieres A. J., Tuñón-Blanco P., Electroanalysis. 17, 1555 (2005)
- Kilele, J. C., Chokkareddy, R., Rono, N., Redhi, G. G., Taiwan, J. Inst. Chem.Eng. 111, 228 (2020)
- Chokkareddy, R., . Redhi, G. G, Thangavel, K., Anal. Sci. 37, 1265 (2021)
- Chokkareddy, R., Thondavada, N., Kabane, B., Redhi, G. G., J. Iran. Chem. Soc.18, 621 (2021)
- Chokkareddy, R., Kanchi, S., Inamuddin, Sci. Rep. 10, 2 (2020)
- Shetti, N. P., Malode, S. J., Nayak, D. S., Aminabhavi, T. M., Reddy, K. R.,Microchem. J. 150, 104124 (2019)
- Purohit, B., Vernekar, P. R., Shetti, N. P., Chandra, P., Sensors Int. 1, 100040(2020)
- Kilele, J. C., Chokkareddy, R., Redhi, G. G., Microchem. J. 164, 106012 (2021)
- Shetti, N. P., S. Malode, J., Vernekarg, P. R., Nayak, D. S., Shetty, N. S., Reddy, K. R., Shukla, S. S., Aminabhavi, T. M., Microchem. J. 149, 103976 (2019)
- Shetti, N. P., Malode, S. J., Ilager, D., Raghava Reddy, K., Shukla, S. S., Aminabhavi, T. M., Electroanalysis. 31, 1040 (2019)
- Moon, J. M., Thapliyal, N., Hussain, K. K., Goyal, R. N., Shim, Y. B., Biosens.Bioelectron. 102, 540 (2018)
- Rajesh, T. Ahuja, Kumar, D., Sensors Actuators, B Chem. 136, 275 (2009)
- Ozkan, S. Z., Bondarenko, G. N., Karpacheva, G. P., Polym. Sci. Ser. B 52, 263.
- Abo, El-Maali, N., Bio electrochemistr. 64, 99 (2004)
- Kilele, J. C., Chokkareddy, R., Rono, N., Redh,i G. G., J. Taiwan Inst. Chem.Eng. 111, 228 (2020)
- Chokkareddy, R., Redhi, G. G., Thangavel, K., Anal. Sci. 37, 1265 (2021)
- Chokkareddy, R., Thondavada, N., Kabane, B., Redhi, G. G., J. Iran. Chem. Soc.18, 621 (2021)
- Chokkareddy, R., Kanchi, S., Inamuddin, Sci. Rep. 10, 2 (2020)
- Shetti, N. P., Malode, S. J., Nayak, D. S., Aminabhavi, T. M., Reddy, K. R.,Microchem. J. 150, 104124 (2019)
- Purohit, B., Vernekar, P. R., Shetti, N. P., Chandra, P., Sensors Int. 1, 100040(2020)
- Kilele, J. C., Chokkareddy, R., Redhi, G. G., Microchem. J. 164, 106012 (2021)
- Shetti, N. P., Malode, S. J., Vernekarg, P. R., Nayak, D. S., Shetty, N. S., Reddy, K. R., Shukla, S. S., Aminabhavi, T. M., Microchem. J. 149, 103976 (2019)
- Shetti, N. P., Malode, S. J., Ilager, D., Raghava Reddy, K., Shukla, S. S., Aminabhavi, T. M., Electroanalysis. 31, 1040 (2019)
- Srivastava, A.K., Srivastava, K., Prasad, J., Synthesis, characterization, biological (in vitro) activity and electrochemical studies of mixed-ligand copper (II) and cobalt (II) complexes with picolinic acid and imides, Chem. Data Collect. 31, 100620 (2021)
- Topală, T., Bodoki, A. , Oprean, L., Oprean,R., Experimental Techniques Tmployed in the study of Metal Complexes-DNA, Interactions , Farmacia, 62 (6) 2014
- Scott Mdndone, J., Vikse, L. K., Assigning the ESI mass spectra of organometallic and coordination compounds, J. Mass Spectrum, 54, 466-479 (2019)
- Moon, J. M., Thapliyal, N., Hussain, K. K., Goyal, R. N., Shim, Y. B., Biosens. Bioelectron. 102, 540 (2018)
- Rajesh Ahuja, T., Kumar, D., Sensors Actuators, B Chem. 136, 275 (2009)
- Shaker, A.M., Nassr, L.A.E., Adam, M.S.S., I.M.A. Mohamed, Hydrophilicity and acid hydrolysis of water-soluble antibacterial iron (II) Schiff base complexes in binary aqueous solvents, Rus. J. Gen. Chem. 83 2460–2464 (2013)
- Boghaei, D.M., Askarizadeh, E., Bezaatpour, A., Synthesis, characterization, spectroscopic and thermodynamic studies of charge transfer interaction of a new water soluble cobalt (II) Schiff base complex with imidazole derivatives. Spectrochim. Acta A Mol. Biomol. Spectrosc. 69 624–628 (2008)
- Bard, A.J., Faulkner, L.R., Electrochemical Methods: Fundamental and Applications, Johns Wiley and Sons, Inc., New York. 429 (1980), Chap. 2.
- Srivastava, A. K., Electrochemical and spectral investigation of copper (II) complexes with various hydroxynicotinic acids in dimethylsulfoxide,Chemical Data Collections. 36, 100789 (2021)
- Srivastava, K., Singh, N., Srivastava, A.K., Prasad, J., Cyclic voltammetric and spectral studies of some mixed-ligands copper(II) complexes involving 2,2′ – bipyridine/1,10-phenanthroline and amino acids, J. Indian Chem. Soc. 86, 139–142 (2009)
- Srivastava, K., Srivastava, A.K., Prasad, J., Electrochemical and spectral studies of binuclear monohydroxo-bridged copper(II) complexes, J. Indian Chem. Soc. 86, 23(2009)
- Da Silva, F., The chelate effect redefined, J. Chem. Educ. 60, 390 (1983)

Pingback: tetracycline 500mg tablet
Pingback: prevacid generic otc
Pingback: nitrofurantoin mono
Pingback: lasix generic medication
Pingback: sildenafil directions
Pingback: dapoxetine 30mg cvs